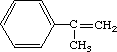

With regard to repeat dose toxicity, one male rat died due to ischuria with urinary calculi in the 1000 mg/kg/day group. Increases in GPT, BUN and K, a decrease in triglyceride were observed in this group. Histopathological examination revealed acidophilic changes in the liver cells, hyaline degeneration of tubular epithelium of kidneys and fatty droplets in adrenal cortex of both sexes, and epithelial hyperplasia of urinary bladder in male rats, and atrophy of thymus in female rats in the 1000 mg/kg/day group. In the 200 mg/kg/day group, similar histopathological changes were observed in the liver, kidneys and thymus. The NOELs for repeat dose toxicity are considered to be 40 mg/kg/day for both sexes. In terms of reproductive/developmental toxicity, the compound exerted effects on the body weight of neonates and the viability index, due to total litter loss in two of eight dams receiving 1000 mg/kg/day. The NOELs for reproductive/developmental toxicity are considered to be 1000 mg/kg/day for parental males, and 200 mg/kg/day for parental females and offspring.
1-Methylethenylbenzene was not mutagenic in Salmonella typhimurium TA100, TA98, TA1535, TA1537 and Escherichia coli WP2 uvrA in the absence or presence of an exogenous metabolic activation system.
Neither structural chromosomal aberrations nor polyploidy were induced in CHL/IU cells up to the concentration giving 50% cell growth inhibition, in the absence or presence of an exogenous metabolic activation system.
| Purity | : | 99.6% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | OECD Combined Repeat Dose and Reproductive/Developmental Toxicity Screening Test |
| ””Route | : | Oral (gavage) |
| ””Dosage | : | 0 (Vehicle), 40, 200, 1000 mg/kg/day |
| ””Number of animals | : | Males, 10; females, 10/group |
| ””Vehicle | : | Olive oil |
| ””Administration period | : | Males, 43 days Females, from 14 days before mating to day 3 of lactation |
| ””Terminal kill | : | Males, day 44 Females, day 4 of lactation |
| GLP | : | Yes |
””Test results:
| Purity | : | 99.6% |
| Test species/strain | : | S.typhimurium TA100, TA1535, TA98, TA1537 E. coli WP2 uvrA |
| Test method | : | Guideline for Screening Mutagenicity Testing of Chemicals (Japan) |
| Procedures | : | Plate incorporation method |
| ””Solvent | : | DMSO |
| ””Positive controls | : | -S9 Mix, AF-2 (TA100, WP2, TA98), sodium azide (TA1535) and 9-aminoacridine (TA1537) +S9 Mix, 2-aminoanthracene (all strains) |
| ””Doses | : | 0, 12.5, 25, 50, 100, 200 and 400 ¦Ģg/plate |
| ””S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
| + | ? | - | |
| without metabolic activation: | [ ] | [ ] | [*] |
| with metabolic activation: | [ ] | [ ] | [*] |
E. coli WP2 uvrA
| + | ? | - | |
| without metabolic activation: | [ ] | [ ] | [*] |
| with metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99.6% |
| Type of cell used | : | Chinese hamster lung (CHL/IU) cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ””Solvent | : | Dimethyl sulfoxide |
| ””Positive controls | : | -S9, Mitomycin C +S9, Cyclophosphamide |
| ””Doses | : | -S9 (continuous treatment): 0, 0.04, 0.09, 0.17 mg/ml -S9 (short-term treatment): 0, 0.04, 0.09, 0.17 mg/ml +S9 (short-term treatment): 0, 0.06, 0.12, 0.23 mg/ml |
| ””S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
Lowest concentration producing cytogenetic effects in vitro:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| without metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| with metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| 1) | The tests were performed by the Mitsubishi Chemical Safety Institute Ltd., 14 Sunayama, Hasaki-machi, Kashima-gun, Ibaraki, 314-02, Japan. Tel +81-46-2871 Fax +81-46-2874 |
| 2) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257, Japan. Tel +81-463-82-4751 Fax +81-463-82-9627 |