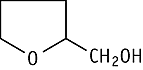

Tetrahydrofurfuryl alcohol was studied for oral toxicity in rats in a 28-day repeated dose toxicity test at doses of 0, 10, 40, 150 and 600 mg/kg/day.
With 600 mg/kg, signs of increased locomotor activity followed by decreased locomotor activity and adoption of a prone position in males and females, and decreases in grip strenth of the hindlimb, body weight gain and food consumption in males were observed. Hematological and blood biochemical examinations revealed decreases in MCH, MCHC, leukocyte and platelet counts and levels of ALP, total protein, albumin, total bilirubin and calcium in males and females, reticulocyte count and levels of LDH, triglyceride and sodium in males, and hemoglobin concentration in females. Prolongation of the prothrombin time in males and females and an increase in BUN level in males were also observed. There were decreases in absolute and relative thymus weights in males and females. Decreases in absolute and relative pituitary weights and an increase in relative kidney weight were also observed in females. Histopathological examination revealed atrophy of the thymus in males and females.
Additionally, in males, atrophy of the red pulp with decreased extramedullary hematopoiesis and inflammation of the capsule in the spleen and necrosis of the seminiferous tubular epithelium in the testes were observed. With 150 mg/kg, signs of increased locomotor activity and decrease in relative pituitary weights in females and decrease in total protein level and necrosis of the seminiferous tubular epithelium in the testes in males were observed. The NOELs are considered to be 40 mg/kg/day for males and females.
Tetrahydrofurfuryl alcohol was studied for oral toxicity in rats in the OECD preliminary reproduction toxicity screening test at doses of 0, 15, 50, 150 and 500 mg/kg/day.
With regard to repeated toxicity, signs of increased locomotor activity or increased locomotor activity followed by decreased locomotor activity was observed in males and females of the 150 and 500 mg/kg groups. Suppression of body weight gain and decrease in food consumption were also observed in both sexes of the same dose groups. The relative kidney weights were increased in females of the 150 mg/kg group. The relative weights of the thymus, testes and epididymides were decreased in males of the 500 mg/kg group. Histopathological examination revealed atrophy of the thymus in both sexes and atrophy of the seminiferous tubules with hyperplasia of the interstitial cells in the testes and decreased intraluminal sperm in the epididymides of the 500 mg/kg group. Atrophy of the red pulp with decreased extramedullary hematopoiesis in the spleen in the 150 and 500 mg/kg groups, and inflammation of the spleen capsules in the 500 mg/kg group were also observed in both sexes. The NOELs for repeated dose toxicity are considered to be 50 mg/kg/day for parental animals of both sexes.
With regard to reproductive/developmental toxicity, the test substance showed effects on reproductive organs of male parents at the dose of 500 mg/kg as described above. Tendencies for prolongation of estrous cycle and gestation length and lowering of the gestation index in female parents, and remarkable decreases in numbers of born pups and live pups on day 0 of lactation and indices for delivery, live birth and viability were observed in the 150 mg/kg group. In the 500 mg/kg group, no females could deliver because of early resorption in the uterus. The NOELs for reproductive/developmental toxicity are considered to be 150 mg/kg/day for males, and 50 mg/kg/day for females and offsprings.
Tetrahydrofurfuryl alcohol was not mutagenic in Salmonella typhimurium TA100, TA1535, TA98, TA1537 or Escherichia coli WP2 uvrA/pKM101, with or without an exogenous metabolic activation system.
Tetrahydrofurfuryl alcohol did not induce structural chromosomal aberrations or polyploidy in CHL/IU cells with or without exogenous metabolic activation system.
| Purity | : | 99.5 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 423 |
| ””Route | : | Oral (Gavage) |
| ””Dosage | : | 0 (Vehicle), 2000 mg/kg |
| ””Number of animals | : | Females, 6 |
| ””Vehicle | : | Distilled water |
| GLP | : | Yes |
””Test results:
The acute toxicity was classified on category 5 in the GHS.
| Purity | : | 99.5 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | Guideline for the 28-Day Repeated Dose Toxicity Test in Mammalian Species (Chemical Substances Control Law of Japan) |
| ””Route | : | Oral (Gavage) |
| ””Dosage | : | 0 (Vehicle), 10, 40, 150, 600 mg/kg |
| ””Number of animals/group | : | Males, 5; females, 5 |
| ””Vehicle | : | Distilled water |
| ””Administration period | : | Males and females, 28 days |
| ””Terminal killing | : | Males and females , days 29 or 43 |
| GLP | : | Yes |
””Test results:
Histopathological examination of testes demonstrated a tendency for increase in severity of changes at the end of the 14-day recovery period. There was a decrease in the ratio of pachytene spermatocyte counts to Sertoli cell counts in addition to that of spermatid to Sertoli cell counts. The other changes observed during or at the end of the administration period showed a tendency for recovery or complete recovery.
In conclusion, tetrahydrofurfuryl alcohol produced effects on the testes, thymus, spleen and kidneys in addition to hematological and blood biochemical effects. The NOEL is considered to be 40 mg/kg/day for males and females.
| Purity | : | 99.5 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 421 |
| ””Route | : | Oral (Gavage) |
| ””Dosage | : | 0 (Vehicle), 15, 50, 150, 500 mg/kg/day |
| ””Number of animals/group | : | Males, 12 ; females, 12 |
| ””Vehicle | : | Distilled water |
| ””Administration period | : | Males, 47 days Females, from 14 days before mating to day 4 of lactation |
| ””Terminal killing | : | Males, day 48 Females, day 5 of lactation |
| GLP | : | Yes |
””Test results:
Signs of increased locomotor activity or increased locomotor activity followed by decreased locomotor activity were observed in males and females of the 150 and 500 mg/kg groups. Suppression of body weight gain and decreased food consumption were also noted in both sexes of the same dose groups. The relative kidney weights were increased in females of the 150 mg/kg group. The relative weights of the thymus, testes and epididymides were decreased in males of the 500 mg/kg group. Gross examination revealed small thymi in both sexes and small testes and epididymes in males of the 500 mg/kg group. Spleens with rough surface, white spots and/or white areas were also observed in both sexes of the 150 and 500 mg/kg groups. Histopathologically, there were atrophy of the thymus in both sexes and atrophy of the seminiferous tubule with hyperplasia of the interstitial cell in the testes and decreased intraluminal sperm with cell debris in the epididymides of the 500 mg/kg group. Atrophy of the red pulp with decreased extramedullary hematopoiesis in the spleen in the 150 and 500 mg/kg groups, and inflammation of the spleen capsule in the 500 mg/kg group were also observed in both sexes.
The NOELs for repeated dose toxicity are considered to be 50 mg/kg/day for parental animals of both sexes.
<Reproductive and developmental toxicity>
The test substance showed effects on reproductive organs of male parents at a dose of 500 mg/kg as described above. Tendencies for prolongation of estrous cycle and gestation length and lowering of the gestation index in female parents, and remarkable decreases in numbers of born pups and live pups on day 0 of lactation and indexes for delivery, live birth and viability were observed in the 150 mg/kg group. In the 500 mg/kg group, none of the females could deliver because of early resorption in the uterus.
The NOELs for reproductive/developmental toxicity are considered to be 150 mg/kg/day for males, and 50 mg/kg/day for females and offsprings.
| Purity | : | 99.5 % |
| Test species/strain | : | Salmonella typhimurium TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA/pKM101 |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 471 |
| ””Procedures | : | Pre-incubation method |
| ””Vehicle | : | Water for injection |
| ””Positive controls | : | -S9 mix; 2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide (TA100, TA98), Sodium azide (TA1535), 9-Aminoacridine hydrochloride (TA1537) and N-Ethyl-N'-nitro-N-nitro-soguanidine (WP2 uvrA/pKM101) +S9 mix; 2-Aminoanthracene (all strains) |
| ””Dosage | : | -S9 mix; 0, 313, 625, 1250, 2500, 5000 ¦Ģg/plate (all strains) +S9 mix; 0, 313, 625, 1250, 2500, 5000 ¦Ģg/plate (all strains) |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
””Test results:
Genetic effects:
Salmonella typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
Escherichia coli WP2 uvrA/pKM101
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99.5 % |
| Type of cell used | : | Chinese hamster CHL/IU cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 473 |
| ””Vehicle | : | Isotonic sodium chloride solution |
| ””Positive controls | : | -S9 mix; Mitomycin C +S9 mix; Benzo[a]pyrene |
| ””Dosage | : | -S9 mix(6-hr short-term treatment); 257.5, 515, 1030 ¦Ģg/mL +S9 mix(6-hr short-term treatment); 257.5, 515, 1030 ¦Ģg/mL -S9 mix(24-hr continuous treatment); 257.5, 515, 1030 ¦Ģg/mL |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
””Test results:
Genetic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| 1) | The tests were performed by the Research Institute for Animal Science in Biochemistry and Toxicology, 3-7-11 Hashimotodai, Sagamihara-shi, Kanagawa, 229-1132, Japan, Tel +81-42-762-2775 Fax +81-42-762-7979 |
| 2) | The tests were performed by the Mitsubishi Chemical Safety Institute Ltd., 14 Sunayama, Hasaki-machi, Kashima-gun, Ibaraki, 314-0255, Japan. Tel +81-479-46-2871 Fax +81-479-46-2874 |