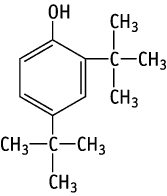

2,4-Di-tert-butylphenol was studied for oral toxicity in rats in a single dose toxicity test at doses of 0, 819, 1024, 1280, 1600 and 2000 mg/kg for both sexes. The single dose oral toxicity test revealed approximate LD50 values of ca. 2000 mg/kg for males and 1762.4 mg/kg for females.
2,4-Di-tert-butylphenol was studied for oral toxicity in Crj:CD(SD)IGS rats in a 28-day repeat dose toxicity test at doses of 0, 5, 20, 75 and 300 mg/kg.
No deaths occurred of either males or females. Salivation was observed in the males and females of the 300 mg/kg group. No effects related to the test substance were observed in terms of body weight or food consumption. Urinalysis showed increase in urine volume, decreases in osmotic pressure and specific gravity, and a pale yellow color in the males and females of the 300 mg/kg group. Hematological examination showed decreases in hemoglobin and hematocrit, and increase in the proportion of segmented neutrophils in the females of the 300 mg/kg group. Further, prolongation of prothrombin time and activated partial thromboplastin time in the males of the 300 mg/kg group was detected. Blood chemical examination showed increases in total cholesterol and phospholipid in the females of the 75 and 300 mg/kg groups. Macroscopically, enlargement of the kidneys was observed in males of the 300 mg/kg group. Light gray spots on the kidneys were observed in females of the 300 mg/kg group. Liver weights were increased in females of the 75 mg/kg group and in males and females of the 300 mg/kg group. Histopathologically, centrilobular hypertrophy of hepatocytes, basophilic change of renal tubules, and granular and proteinaceous casts in the kidneys were observed in the males and females of the 300 mg/kg group. Cellular infiltration (neutrophils), dilatation of the distal tubules and/or hypertrophy of the proximal tubules were also observed in the animals with lesions in the kidney at necropsy. In the recovery test, these changes observed during the administration period demonstrated recovery. The NOELs are considered to be 75 mg/kg/day for males and 20 mg/kg/day for females.
Genotoxicity of 2,4-di-tert-butylphenol was studied by reverse mutation test in bacteria and by chromosomal aberration test in cultured Chinese hamster lung (CHL/IU) cells.
2,4-Di-tert-butylphenol was not mutagenic in Salmonella typhimurium TA100, TA1535, TA98, TA1537 and Escherichia coli WP2 uvrA, with or without an exogenous metabolic activation system.
2,4-Di-tert-butylphenol induced structural chromosomal aberrations in CHL/IU cells after short-term treatment with an exogenous metabolic activation system.
| Purity | : | 99.67 % |
| Test species/strains | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 401 |
| ””Route | : | Oral (gavage) |
| ””Doses | : | 0(vehicle), 819, 1024, 1280, 1600, 2000 mg/kg |
| ””Number of animals/group | : | Males, 5; females, 5 |
| ””Vehicle | : | Corn oil |
| GLP | : | Yes |
””Test results:
| Purity | : | 99.67 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | Guideline for 28-Day Repeated Dose Toxicity Test in Mammalian Species (Chemical Substances Control Law of Japan) |
| ””Route | : | Oral(gavage) |
| ””Dosage | : | 0(vehicle), 5, 20, 75, 300 mg/kg/day |
| ””Number of animals/group | : | Males, 6 or 12(0, 300 mg/kg) |
| ””Vehicle | : | Corn oil |
| ””Administration period | : | Males and females, 28 days |
| ””Terminal kill | : | Males and females, on days 29 and 43(0, 300 mg/kg) |
| GLP | : | Yes |
””Test results:
| Purity | : | 99.67% |
| Test species/strains | : | Salmonella typhimurium TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals(Chemical Substances Control Law of Japan) and OECD Test Guideline 471 |
| ””Procedures | : | Pre-incubation method |
| ””Solvent | : | DMSO |
| ””Positive controls | : | -S9 mix; 2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide (TA100, TA98, WP2 uvrA), Sodium azide (TA1535) and
9-Aminoacridine (TA1537) +S9 mix; 2-Aminoanthracene (all strains) |
| ””Doses | : | -S9 mix; 0, 0.781, 1.56, 3.13, 6.25, 12.5, 25 ¦Ģg/plate(TA100, TA1535, TA1537) -S9 mix; 0, 1.56, 3.13, 6.25, 12.5, 25, 50 ¦Ģg/plate(TA98) -S9 mix; 0, 3.13, 6.25, 12.5, 25, 50, 100 ¦Ģg/plate(WP2 uvrA) +S9 mix; 0, 3.91, 7.81, 15.6, 31.3, 62.5, 125 ¦Ģg/plate(WP2 uvrA) +S9 mix; 0, 7.81, 15.6, 31.3, 62.5, 125, 250 ¦Ģg/plate(TA1535, TA1537) +S9 mix; 0, 15.6, 31.3, 62.5, 125, 250, 500 ¦Ģg/plate(TA100, TA98) |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
””Test results:
Genetic effects:
Salmonella typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| ””Without metabolic activation: | [ ] | [ ] | [*] |
| ””With metabolic activation: | [ ] | [ ] | [*] |
Escherichia coli WP2 uvrA
| + | ? | - | |
| ””Without metabolic activation: | [ ] | [ ] | [*] |
| ””With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99.67 % |
| Type of cell used | : | Chinese hamster lung (CHL/IU) cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals(Chemical Substances Control Law of Japan) and OECD Test Guideline 473 |
| ””Solvent | : | DMSO |
| ””Positive controls | : | -S9 mix; 1-Methyl-3-nitro-1-nitrosoguanidine +S9 mix; 3,4-Benzo[a]pyrene |
| ””Doses | : | -S9 mix(6 hr short-term treatment); 0, 10, 20, 30, 40, 50, 60 ¦Ģg/mL +S9 mix(6 hr short-term treatment); 0, 1.25, 2.5, 5, 7.5, 10 ¦Ģg/mL |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
””Test results:
| Lowest concentration producing cytogenetic effects in vitro: | ||
| ””With metabolic activation (6 hr short-term treatment) | : | 7.5 ¦Ģg/mL(clastogenicity) |
Genetic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| ””Without metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| ””With metabolic activation: | [*] | [ ] | [ ] | [ ] | [ ] | [*] |
| 1) | The tests were performed by the Safety Assessment Laboratory, Panapharm Laboratories Co., Ltd., 1285 Kurisaki-machi, Uto-shi, Kumamoto, 869-0425, Japan Tel +81-964-23-5111 Fax +81-964-23-2282 |
| 2) | The tests were performed by the Research Institute for Animal Science in Biochemistry and Toxicology, 3-7-11 Hashimotodai, Sagamihara-shi, Kanagawa 229-1132, Japan. Tel +81-42-762-2775 Fax +81-42-762-7979 |