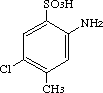

2-Amino-5-chloro-4-methylbenzenesufonic acid was studied for oral toxicity in rats in a single dose toxicity test at a dose of 2000 mg and in a 28-day repeat dose toxicity test at doses of 0, 100, 300 and 1000 mg/kg/day.
2-Amino-5-chloro-4-methylbenzenesufonic acid was also tested for mutagenicity with assays for reverse mutation in bacteria and chromosomal aberrations in cultured Chinese hamster (CHL) cells.
The single dose oral toxicity test revealed LD50s of more than 2000 mg/kg. 2-Amino-5-chloro-4-methylbenzenesufonic acid did not show any toxic effects in the repeat dose study. The NOEL for repeat dose toxicity was 1000 mg/kg/day.
2-Amino-5-chloro-4-methylbenzenesufonic acid was not mutagenic for S. typhimurium TA100, TA1535, TA98, TA1537 and E. coli WP2 uvrA with or without exogenous metabolic activation up to 5000 ¶Őg/plate. However, this chemical induced chromosomal aberrations with or without exogenous metabolic activation at concentrations of 2.5 and 2.0 mg/ml, respectively. The chemical did not induce polyploidy under the test conditions applied.
| Purity | : | > 99.5% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | OECD Test Guideline 401 |
| °°Doses | : | 300, 1000, 2000 mg/kg |
| °°Number of animals | : | Male, 5; Female, 5/group |
| GLP | : | Yes |
| Test results | : | LD50: Male > 2000 mg/kg, Female > 2000 mg/kg |
| Purity | : | > 99.5 % |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | Guidelines for 28-Day Repeat Dose Toxicity Test of Chemicals (Japan) |
| °°Route | : | Oral (gavage) |
| °°Doses | : | 0 (vehicle), 100, 300, 1000 mg/kg/day |
| °°Number of animals | : | Male, 5; Female, 5/group |
| °°Vehicle | : | 0.5% CMC Na aqueous solution |
| °°Administration period | : | Male and Female, 28 days |
| °°Terminal kill | : | Male and Female, days 29 or 43 |
| GLP | : | Yes |
On hematological and blood chemical analyses, there were no differences found from the controls. On urinalysis, positive urobilinogen and pH decrease were seen in both sexes. Additional studies revealed that these changes may not have been caused by the test substance itself, but by chemical interaction of its metabolites with test reagents. There were no organ weight changes in either males or females.
No macroscopic or microscopic abnormalities were seen by pathological examination that could be attributed to treatment with the test substance.
| Purity | : | > 99.5% |
| Test species/strains | : | S. typhimurium TA100, TA1535, TA98, TA1537 E.coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| °°Procedure | : | Plate method |
| °°Solvent | : | DMSO |
| °°Positive controls | : | -S9, AF-2 (TA100,WP2 uvrA,TA98), sodium azide (TA1535) and 9-aminoacridine (TA1537) +S9, 2-aminoanthracene (all strains) |
| °°Doses | : | 0, 156.3, 312.5, 625, 1250, 2500, 5000 ¶Őg/plate |
| °°S-9 | : | Rat liver, induced by phenobarbital and 5,6-benzoflavone |
| °°Plates/test | : | 3 |
| °°Number of replicates | : | 2 |
| GLP | : | Yes |
Genotoxic effects:
S. typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| with metabolic activation: | [ ] | [ ] | [*] |
| without metabolic activation: | [ ] | [ ] | [*] |
E. coli WP2 uvrA
| + | ? | - | |
| with metabolic activation: | [ ] | [ ] | [*] |
| without metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | > 99.5% |
| Type of cell used | : | Chinese hamster CHL cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| °°Vehicle | : | 0.5% CMC Na aqueous solution |
| °°Positive controls | : | -S9, Mitomycin C +S9, Cyclophosphamide |
| °°Doses | : | -S9: 0, 0.5, 1, 2 mg/ml +S9: 0, 0.55, 1.1, 2.2 mg/ml |
| °°S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavon |
| °°Plates/test | : | 2 |
| °°GLP | : | Yes |
Lowest concentration producing cytogenetic effects in vitro:
with metabolic activation: > 2.2 mg/ml
without metabolic activation: > 2 mg/ml
Genotoxic effects:
| + | ? | - | |
| with metabolic activation: | [*] | [ ] | [ ] |
| without metabolic activation: | [*] | [ ] | [ ] |
| 1) | The tests were performed by the Biosafety Research Center, Foods, Drugs and Pesticides (An-pyo Center), 582-2 Shioshinden Arahama, Fukude-cho, Iwata-gun, Shizuoka, 437-12, Japan. Tel 81-538-58-1266 Fax 81-538-58-1393 |
| 2) | The test were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257, Japan. Tel 81-463-82-4751 Fax 81-463-82-9627 |