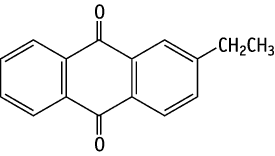

2-Ethylanthraquinone was studied for oral toxicity in rats in a 28-day repeat dose toxicity test at doses of 0, 10, 50 and 250 mg/kg. No deaths were observed in either sex. The density of erythrocytes was decreased in males given 10 mg/kg or more and in females given 250 mg/kg. In parallel, decreased hematocrit values, and increases in MCHC, the proportion of reticulocytes, the level of total bilirubin, hemosiderosis in the spleen and extramedullary hematopoiesis were found mainly in the 250 mg/kg group. Moreover, the animals of both sexes given 50 mg/kg and more demonstrated centrilobular hypertrophy of hepatocytes, together with anisonucleosis in severe cases. Thus, the NOELs for the 28-day repeat dose oral toxicity test of 2-ethylanthraquinone are considered to be less than 10 mg/kg/day for males and 10 mg/kg/day for females.
Reverse mutation assays using microorganisms (Salmonella typhimurium, Escherichia coli) were conducted to assess the potential of 2-ethylanthraquinone to induce gene mutations. 2-Ethylanthraquinone did not induce gene mutations in bacteria under the conditions of this study.
In vitro chromosomal aberration tests using cultured cells (CHL/IU) were conducted to assess the potential of 2-ethylanthraquinone to induce chromosomal aberrations. 2-Ethylanthraquinone induced chromosomal aberrations in cultured cells under the conditions of this study.
| Purity | : | 99.16 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 401 |
| ””Route | : | Oral(gavage) |
| ””Doses | : | Males, 0(vehicle), 2000 mg/kg Females, 2000 mg/kg |
| ””Number of animals/group | : | Males, 5; females, 5 |
| ””Vehicle | : | 0.5 w/v% CMC Na containing 0.1 w/v% polysorbate 80 |
| GLP | : | Yes |
””Test results:
The LD50 value was concluded to be more than 2000 mg/kg for both sexes.
| Purity | : | 99.16 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | Guideline for 28-Day Repeated Dose Toxicity Test in Mammalian Species (Chemical Substances Control Law of Japan) |
| ””Route | : | Oral(gavage) |
| ””Doses | : | 0(vehicle), 10, 50, 250 mg/kg/day |
| ””Number of animals/group | : | Males, 10; females, 10(0, 250 mg/kg) Males, 5; females, 5(10, 50 mg/kg) |
| ””Vehicle | : | 0.5 % CMC Na containing 0.1 % polysorbate 80 |
| ””Administration period | : | Males and females, 28 days |
| ””Terminal kill | : | Males and females, on days 29 and 43 |
| GLP | : | Yes |
””Test results:
Thus, NOELs for the 28-day repeat dose toxicity are considered to be less than 10 mg/kg/day for males and 10 mg/kg/day for females.
| Purity | : | 99.16 % |
| Test species/strains | : | Salmonella typhimurium TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals(Chemical Substances Control Law of Japan) and OECD Test Guideline 471 |
| ””Procedures | : | Pre-incubation method |
| ””Solvent | : | DMSO |
| ””Positive controls | : | -S9 mix; 2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide (TA100, TA98 and WP2 uvrA), Sodium azide (TA1535) and
9-Aminoacridine hydrochloride (TA1537) +S9 mix; 2-Aminoanthracene (all strains) |
| ””Doses | : | -S9 mix; 0, 156, 313, 625, 1250, 2500, 5000 ¦Ģg/plate(all strains) +S9 mix; 0, 2.44, 4.88, 9.77, 19.5, 39.1, 78.1, 156 ¦Ģg/plate(TA100) +S9 mix; 0, 156, 313, 625, 1250, 2500, 5000 ¦Ģg/plate(TA1535, TA98, TA1537) +S9 mix; 0, 39.1, 78.1, 156, 313, 625, 1250, 2500, 5000 ¦Ģg/plate(WP2 uvrA) +S9 mix(confirmative test, 1st); 0, 2.44, 4.88, 9.77, 19.5, 39.1, 78.1, 156, 313, 625, 1250, 2500, 5000 ¦Ģg/plate(TA100) +S9 mix(confirmative test, 2nd); 0, 156, 313, 625, 1250, 2500, 5000 ¦Ģg/plate(TA100) |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
””Test results:
Genetic effects:
Salmonella typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| ””Without metabolic activation: | [ ] | [ ] | [*] |
| ””With metabolic activation: | [ ] | [ ] | [*] |
Escherichia coli WP2 uvrA
| + | ? | - | |
| ””Without metabolic activation: | [ ] | [ ] | [*] |
| ””With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99.16 % |
| Type of cell used | : | Chinese hamster lung (CHL/IU) cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals(Chemical Substances Control Law of Japan) and OECD Test Guideline 473 |
| ””Solvent | : | DMSO |
| ””Positive controls | : | -S9 mix; Mitomycin C +S9 mix; Cyclophosphamide |
| ””Doses | : | -S9 mix(short-term treatment); 0, 40.0, 80.0, 160, 320, 640 ¦Ģg/mL +S9 mix(short-term treatment); 0, 591, 1181, 2362 ¦Ģg/mL +S9 mix(short-term treatment, confirmative test); 0, 37.5, 75.0, 150, 300, 600 ¦Ģg/mL |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
””Test results:
Genetic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| ””Without metabolic activation: | [ ] | [ ] | [*] | [*] | [ ] | [ ] |
| ””With metabolic activation: | [ ] | [*] | [ ] | [ ] | [ ] | [*] |
| 1) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257-8523, Japan. Tel +81-463-82-4751 Fax +81-463-82-9627 |
| 2) | The tests were performed by the Biosafety Research Center, Foods, Drugs and Pesticides(An-pyo Center), 582-2 Shioshinden, Fukude-cho, Iwata-gun, Shizuoka, 437-1213, Japan. Tel +81-538-58-1266 Fax +81-538-58-1393 |