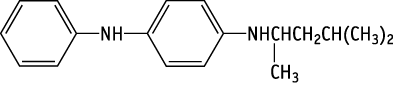

N-(1,3-Dimethylbutyl)-N'-phenyl-p-phenylenediamine was studied for oral toxicity in rats in a reproduction/developmental toxicity screening test at doses of 0, 6, 25 and 100 mg/kg/day.
In the test, 1 dam of the 100 mg/kg group died on day 23 of gestation. Salivation was observed in the 25 mg/kg or more groups in males and in the 100 mg/kg group in females. The test substance had no effect on body weight and food consumption in any treatment group.
Increases in liver weights were observed in the 25 mg/kg or more groups, in both sexes. As a gross finding, enlargement of the liver was observed in the 100 mg/kg group in both sexes and vacuolar degeneration of the liver was evident in the 25 mg/kg or more groups in males on histological examination. However, there were no adverse effects of test substance on the reproductive organs in either sex in terms of histological findings.
The NOELs for repeat dose toxicity are considered to be 6 mg/kg/day for both sexes.
With regard to reproductive/developmental toxicity, no adverse effects were observed in terms of the estrus cycle, copulation and fertility results or findings for delivery.
No abnormal findings related to the test substance were noted on external examination, or for clinical signs, growth or necropsy findings for the offspring.
The NOELs for reproductive and developmental toxicity are considered to be 100 mg/kg/day for both parental animals and offspring.
| Purity | : | 99.40 wt% |
| Test species/strains | : | Rat/Crj:CD(SD) |
| Test method | : | OECD Test Guideline 401 |
| ””Route | : | Oral (gavage) |
| ””Doses | : | 0(vehicle), 6, 25, 100 mg/kg/day |
| ””Number of animals/group | : | Males, 12; females, 12 |
| ””Vehicle | : | Corn oil |
| ””Administration period | : | Males, 48 days Females, from 14 days before mating to day 3 of lactation |
| ””Terminal kill | : | Males, day 48 Females, day 4 of lactation |
| GLP | : | Yes |
””Test results:
<Repeat dose toxicity>
<Reproductive and Developmental toxicity>
| 1) | The test was performed by the Biosafety Research Center, Foods, Drugs and Pesticides(An-pyo Center), 582-2 Shioshinden Arahama, Fukude-cho, Iwata-gun, Shizuoka, 437-1213, Japan. Tel +81-538-58-1266 Fax +81-538-58-1393 |