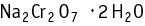
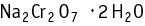
Chromic acid disodium salt dihydrate was studied for oral toxicity in rats in an OECD combined repeated dose and reproductive/developmental toxicity screening test at doses of 0, 1, 6, and 30 mg/kg.
No deaths occurred in any of the treated groups. Salivation after dosing was observed in both sexes of the 30 mg/kg group. Decrease in body weight gain and food consumption on Day 4 of lactation was observed in females of the 30 mg/kg group. In addition, total litter loss was observed in one female of the 30 mg/kg group. Hematological examination revealed decrease in MCH and MCHC in both sexes of the 30 mg/kg group, with increase in reticulocytes in males, and decrease in hemoglobin in females. Histopathologically, the effects of test substance were erosion and ulceration in the stomach, and changes of renal tubular epithelium in females of the 30 mg/kg group. Blood chemistry examination revealed decrease in total protein in males of the 6 and 30 mg/kg groups, and increase in Cl in males of the 30 mg/kg group. The NOEL for repeated dose toxicity is considered to be 1 mg/kg/day for both sexes.
With regard to reproductive and developmental toxicity, the compound caused extension of gestation length in females of the 30 mg/kg group. It had no effects on other parameters. The NOELs for reproductive and developmental toxicity are considered to be 30 mg/kg/day for males, 6 mg/kg/day for females, and 30 mg/kg/day for offspring.
Genotoxicity of chromic acid disodium salt dihydrate was studied by a reverse mutation test in bacteria. This substance was mutagenic in Salmonella typhimurium TA100, TA98, TA1537 and Escherichia coli WP2 uvrA, with or without an exogenous metabolic activation system.
Genotoxicity of chromic acid disodium salt dihydrate was studied by chromosomal aberration test in cultured Chinese hamster lung (CHL/IU) cells.
Chromic acid disodium salt dihydrate induced structural chromosomal aberrations dose-dependently under the short-term treatment conditions with and without metabolic activation, at doses of 0.0056-0.023 mg/mL and 0.00056-0.0023 mg/mL, respectively.
| Purity | : | 100.07 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 401 |
| ЎЎRoute | : | Oral (gavage) |
| ЎЎDosage | : | 0 (vehicle), 100, 130, 180, 240, 330 mg/kg |
| ЎЎNumber of animals/group | : | Males, 5; females, 5 |
| ЎЎVehicle | : | Water for injection |
| GLP | : | Yes |
ЎЎTest results:
LD50: Males, 252.4 mg/kg; females, 181.0 mg/kg
| Purity | : | 100.07 % |
| Test species/strain | : | Rats/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 422 |
| ЎЎRoute | : | Oral (gavage) |
| ЎЎDosage | : | 0 (vehicle), 1, 6, 30 mg/kg |
| ЎЎNumber of animals/group | Males, 12; females, 12 | |
| ЎЎVehicle | : | Water for injection |
| ЎЎAdministration period | : | Males, 37 days Females, from 14 days before mating to day 4 of lactation |
| ЎЎTerminal killing | : | Males, day 38 Females, day 5 of lactation |
| GLP | : | Yes |
ЎЎTest results:
No deaths occurred in any of the treated groups. Salivation after dosing was observed in of both sexes animals of the 30 mg/kg group. Decrease in body weight gain and food consumption on Day 4 of lactation was observed in females of the 30 mg/kg group. In addition, total litter loss was observed in one female of the 30 mg/kg group. Hematological examination revealed decrease in MCH and MCHC in both sexes, increase in reticulocytes in males, and decrease in hemoglobin in females of the 30 mg/kg group. Histopathologically, erosion and ulceration in the stomach, was evident in both sexes of the 6 and 30 mg/kg groups, and changes of the renal tubular epithelium in females of the 30 mg/kg group. Examination of blood chemistry revealed decrease in total protein in males of the 6 and 30 mg/kg groups, and increase in Cl in males of the 30 mg/kg group.
The NOEL for repeated dose toxicity is considered to be 1 mg/kg/day for both sexes.
<Reproductive and developmental toxicity>
In the 30 mg/kg group, extension of gestation length was observed in female parental animals. The compound had no effects on other reproductive parameters such as the mating index, the fertility index, number of corpora lutea or implantations, the implantation index, the delivery index, the gestation index, parturition or maternal behavior. On examination of neonates, there were no significant differences in number of offspring or live offspring, the sex ratio, the live birth index, the viability index or body weight. No abnormal findings ascribable to the compound were found regarding external features, clinical signs or necropsy of the offspring.
The NOELs for reproductive and developmental toxicity are considered to be 30 mg/kg/day for males, 6 mg/kg/day for females, and 30 mg/kg/day for offspring.
| Purity | : | 100.07 % |
| Test species/strains | : | Salmonella typhimurium TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 471 |
| ЎЎProcedures | : | Pre-incubation method |
| ЎЎVehicle | : | Water for injection |
| ЎЎPositive controls | : | -S9 mix; 2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide (TA100, TA98, WP2 uvrA), Sodium azide (TA1535) and 9-Aminoacridine (TA1537) +S9 mix; 2-Aminoanthracene (five strains) |
| ЎЎDosage | : | -S9 mix; 0, 1.95, 3.91, 7.81, 15.6, 31.3, 62.5 ҰМg/plate (TA100) -S9 mix; 0, 0.977, 1.95, 3.91, 7.81, 15.6, 31.3, 62.5 ҰМg/plate (TA1535, TA98, TA1537) -S9 mix; 0, 7.81, 15.6, 31.3, 62.5, 125, 250 ҰМg/plate (WP2 uvrA) +S9 mix; 0, 3.91, 7.81, 15.6, 31.3, 62.5, 125 ҰМg/plate (TA100, TA1535, TA98, TA1537) +S9 mix; 0, 15.6, 31.3, 62.5, 125, 250, 500 ҰМg/plate (WP2 uvrA) |
| ЎЎS9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ЎЎPlates/test | : | 3 (1 for cytotoxicity test) |
| ЎЎNumber of replicates | : | 2 (plus 1 cytotoxicity test) |
| GLP | : | Yes |
ЎЎTest results:
Genetic effects:
Salmonella typhimurium TA100, TA98
| + | ? | - | |
| Without metabolic activation: | [*] | [ ] | [ ] |
| With metabolic activation: | [*] | [ ] | [ ] |
Salmonella typhimurium TA1535
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
Salmonella typhimurium TA1537
| + | ? | - | |
| Without metabolic activation: | [*] | [ ] | [ ] |
| With metabolic activation: | [ ] | [ ] | [*] |
Escherichia coli WP2 uvrA
| + | ? | - | |
| Without metabolic activation: | [*] | [ ] | [ ] |
| With metabolic activation: | [*] | [ ] | [ ] |
| Purity | : | 100.07 % |
| Type of cell used | : | Chinese hamster lung (CHL/IU) cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 473 |
| ЎЎVehicle | : | Water for injection |
| ЎЎPositive controls | : | -S9 mix; Mitomycin C +S9 mix; Cyclophosphamide |
| ЎЎDosage | : | -S9 mix (short-term treatment); 0, 0.00056, 0.0011, 0.0023 mg/mL +S9 mix (short-term treatment); 0, 0.0056, 0.011, 0.023 mg/mL |
| ЎЎS9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ЎЎPlates/test | : | 2 |
| GLP | : | Yes |
ЎЎTest results:
| Lowest concentration producing cytogenetic effects in vitro | : | ||
| Without metabolic activation (short-term treatment) | : | 0.00056 mg/mL (clastogenicity) | |
| With metabolic activation (short-term treatment) | : | 0.011 mg/mL (clastogenicity) | |
Genotoxic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| Without metabolic activation: | [*] | [ ] | [ ] | [ ] | [ ] | [*] |
| With metabolic activation: | [*] | [ ] | [ ] | [ ] | [ ] | [*] |
| 1) | The test was performed by the Safety Assessment Laboratory, Panapharm Laboratories Co., LTD., 1285 Kurisaki-machi, Uto-shi, Kumamoto, 869-0425, Japan. Tel +81-964-23-5111 Fax +81-964-23-2282 |
| 2) | The test was performed by the Kashima Laboratory, Mitsubishi Chemical Safety Institute Ltd., 14 Sunayama, Hasaki-machi, Kashima-gun, Ibaraki, 314-0255, Japan. Tel +81-479-46-2871 Fax +81-479-46-2874 |
| 3) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257-8523, Japan. Tel +81-463-82-4751 Fax +81-463-82-9627 |