 ) sulfate heptahydrate
) sulfate heptahydrate ) sulfate heptahydrate
) sulfate heptahydrate )МЗПхЯТЪЊ
)МЗПхЯТЪЊ
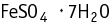
Ferrous sulfate heptahydrate was studied for oral toxicity in rats in an OECD combined repeated dose and reproductive/developmental toxicity screening test at doses of 0, 30, 100, 300, and 1000 mg/kg/day.
Deaths were noted of 1 animal of each sex given 1000 mg/kg. Body weight was low in the males at 1000 mg/kg and tended to be low in the females at 1000 mg/kg in the final stages of pregnancy. Food consumption was transiently low in both sexes at 1000 mg/kg. On urinalysis, a high value for volume and a low value for specific gravity were noted in the males at 1000 mg/kg. In the hematological examination, low values for the red blood cell count and APTT and high values for MCV, MCH, and the reticulocyte count were noted in the males at 1000 mg/kg. In the blood chemical analysis, a high value for inorganic phosphate was noted in the females at 300 mg/kg, low values for total protein, albumin, and Ca and high values for ALT, ІУ-GTP, and A/G in the males at 1000 mg/kg, and high values for ІУ-GTP and inorganic phosphate in the females at 1000 mg/kg. On organ weight measurement, high values for the absolute and relative adrenal weights and the relative liver weights were noted in the males at 1000 mg/kg, and high values for the absolute and relative liver weights were obtained for the females at 1000 mg/kg. On histopathological examination, extramedullary hematopoiesis of the spleen was noted in the males at 300 mg/kg, atrophy of the thymus, inflammatory cell filtration in the glandular submucosa, deposit of yellowish brown pigment in the periportal hepatocytes, deposits of yellowish brown pigment in periportal Kupffer cells, extramedullary hematopoiesis of the spleen, deposits of yellowish brown pigment in the red pulp of the spleen, and basophilic changes in the tubular epithelium of the kidney were noted mainly in the males at 1000 mg/kg. Deposits of yellowish brown pigment in the red pulp of the spleen and of yellowish brown pigment in the periportal hepatocytes were noted in the females at 1000 mg/kg. The NOEL for repeated dose toxicity is considered to be 100 mg/kg/day for both sexes.
Regarding the reproductive and developmental toxicity, no changes attributable to the test substance were noted in parental animals or pups. The NOEL for reproductive performance is considered to be at 1000 mg/kg/day for parental animals and pups.
Ferrous sulfate heptahydrate was not mutagenic in Salmonella typhimurium TA100, TA1535, TA98, TA1537 and Escherichia coli WP2 uvrA/pKM101, with or without an exogenous metabolic activation system.
Ferrous sulfate heptahydrate induced structural chromosomal aberrations in CHL/IU cells after short term treatment with and without an exogenous metabolic activation system. Polyploidy was not induced in any treatment group.
| Purity | : | 91.6 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 401 |
| ЁЁRoute | : | Oral (gavage) |
| ЁЁDosage | : | 0 (vehicle), 250, 500, 1000, 2000 mg/kg |
| ЁЁNumber of animals/group | : | Males, 5; females, 5 |
| ЁЁVehicle | : | Water for injection |
| GLP | : | Yes |
ЁЁTest results:
| Purity | : | 91.1 and 90.6 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 422 |
| ЁЁRoute | : | Oral (gavage) |
| ЁЁDosage | : | 0 (vehicle), 30, 100, 300, 1000 mg/kg |
| ЁЁNumber of animals/group | Males, 12; females, 12 | |
| ЁЁVehicle | : | Water for injection |
| ЁЁAdministration period | : | Males, 49 days Females, from 14 days before mating to day 5 of lactation |
| ЁЁTerminal killing | : | Males, day 50 Females, day 6 of lactation |
| GLP | : | Yes |
ЁЁTest results:
Deaths were noted of 1 animal of each sex at 1000 mg/kg. Body weights were low in the males at 1000 mg/kg and tended to be low in the females at 1000 mg/kg in the final stages of pregnancy. Food consumption was transiently lowered in both sexes at 1000 mg/kg. On urinalysis, a high value for volume and a low value for specific gravity were noted in the males at 1000 mg/kg. In the hematological examination, low values for the red blood cell count and APTT and high values for MCV, MCH, and the reticulocyte count were noted in the males at 1000 mg/kg. In the blood chemical analysis, a high value for inorganic phosphate was noted in the females at 300 mg/kg, low values for total protein, albumin, and Ca and high values for ALT, ІУ-GTP, and A/G in the males at 1000 mg/kg, and high values for ІУ-GTP and inorganic phosphate in the females at 1000 mg/kg. On necropsy, dark red spots in the glandular mucosa and ulcers were noted in the males at 1000 mg/kg. High values for the absolute and relative adrenal weights and the relative liver weights were noted in the males at 1000 mg/kg, and high values for the absolute and relative liver weights were obtained for the females at 1000 mg/kg. On histopathological examination, extramedullary hematopoiesis of the spleen was noted in males at 300 mg/kg. Atrophy of the thymus, ulcers and erosion in the glandular stomach, inflammatory cell filtration in the glandular submucosa, hemorrhage in the stomach, vacuolization in the forestomach epithelium, deposit of yellowish brown pigment in the periportal hepatocytes, deposits of yellowish brown pigment in periportal Kupffer cells, extramedullary hematopoiesis of the spleen, deposits of yellowish brown pigment in the red pulp of the spleen, basophilic changes in the tubular epithelium of the kidney, and increased hematopoiesis of the femoral bone marrow were noted in the males at 1000 mg/kg. Deposits of yellowish brown pigment in the periportal hepatocytes and of yellowish brown pigment in the red pulp of the spleen were noted in the females at 1000 mg/kg.
The NOEL for repeated dose toxicity is considered to be at 100 mg/kg/day for both sexes.
<Reproductive and developmental toxicity>
Regarding the reproductive and developmental toxicity, no changes attributable to the test substance were noted in terms of the number of estrous cases, copulation index, number of days before copulation, fertility index, gestation length, gestation index, delivery conditions, nursing conditions, number of corpora lutea, number of implantation sites, or implantation rate.
The NOEL for reproductive performance of parental animals is considered to be 1000 mg/kg/day for both sexes.
Regarding the pups, no changes attributable to the test substance were noted in terms of the number, number of stillbirths, number of live pups on Day 0 of lactation, sex ratio, delivery index, birth index, live birth index, general signs, number of live pups on Day 4 of lactation, viability index on Day 4 of lactation, external observation, body weight change, or necropsy findings.
The NOEL for pups is considered to be 1000 mg/kg/day.
| Purity | : | 91.6 % |
| Test species/strain | : | Salmonella typhimurium TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA/pKM101 |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 471 |
| ЁЁProcedures | : | Pre-incubation method |
| ЁЁVehicle | : | Water for injection |
| ЁЁPositive controls | : | -S9 mix; 2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide (TA100, TA98), Sodium azide (TA1535), 9-Aminoacridine hydrochloride (TA1537) and N-Ethyl-N'-nitro-N-nitrosoguanidine (WP2 uvrA/pKM101) +S9 mix; 2-Aminoanthracene (five strains) |
| ЁЁDosage | : | -S9 mix; 0, 156, 313, 625, 1250, 2500, 5000 ІЬg/plate (five strains) +S9 mix; 0, 156, 313, 625, 1250, 2500, 5000 ІЬg/plate (five strains) |
| ЁЁS9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ЁЁPlates/test | : | 3 |
| ЁЁNumber of replicates | : | 2 |
| GLP | : | Yes |
ЁЁTest results:
Genetic effects:
Salmonella typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
Escherichia coli WP2 uvrA/pKM101
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 91.6 % |
| Type of cell used | : | Chinese hamster CHL/IU cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 473 |
| ЁЁVehicle | : | Isotonic sodium chloride solution |
| ЁЁPositive controls | : | -S9 mix, Mitomycin C +S9 mix, Benzo[a]pyrene |
| ЁЁDosage | : | -S9 mix (6 hr short-term treatment); 0, 250, 500, 1000, 1500, 2000 ІЬg/mL (main test) -S9 mix (6 hr short-term treatment); 0, 1200, 1300, 1400, 1500, 1600 ІЬg/mL (confirmation test) +S9 mix (6 hr short-term treatment); 0, 125, 250, 500, 1000, 1500 ІЬg/mL (main test) +S9 mix (6 hr short-term treatment); 0, 600, 700, 800, 900, 1000 ІЬg/mL (confirmation test) |
| ЁЁS9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ЁЁPlates/test | : | 2 |
| GLP | : | Yes |
ЁЁTest results:
| Lowest concentration producing cytogenetic effects in vitro | : | ||
| Without metabolic activation (short-term treatment) | : | 1.2 mg/mL (clastogenicity) | |
| With metabolic activation (short-term treatment) | : | 0.6 mg/mL (clastogenicity) | |
Genotoxic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| Without metabolic activation: | [*] | [ ] | [ ] | [ ] | [ ] | [*] |
| With metabolic activation: | [*] | [ ] | [ ] | [ ] | [ ] | [*] |
| 1) | The tests were performed by Nihon Bioresearch Inc., 6-104, Majima, Fukuju-cho, Hashima, Gifu, 501-6251, Japan. Tel +81-58-392-6222 Fax +81-58-392-1284 |
| 2) | The tests were performed by the Mitsubishi Chemical Safety Institute Ltd., 14 Sunayama, Hasaki-machi, Kashima-gun, Ibaraki 314-0255, Japan. Tel +81-479-46-2871 Fax +81-479-46-2874 |