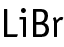
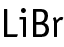
A combined repeated dose and reproductive/developmental toxicity screening test was conducted in rats according to OECD test guideline 422. Stereotyped behavior of rubbing the cage floor with the chin was induced by repeated dosing at 80 mg/kg of lithium bromide in both males and females. Body weight gain was increased in the early period of dosing of 80 mg/kg in the males and females. Lithium bromide treatment did not affect the estrous cycle at doses of 5-80 mg/kg. A few pairs receiving 20 mg/kg and more showed retardation of copulation for 3 estrous cycles or more, but the overall copulation index was not significantly different from that in the control group. Fertility and various gestational indexes were not affected by the treatment. Effects of lithium bromide were not observed in the neonates in any groups. The no observed effect dose level (NOEL) for repeated dose toxicity of lithium bromide in rats is considered to be 5 mg/kg/day in both males and females. The NOEL for reproductive/developmental toxicity is considered to be 5 mg/kg/day in both sexes of animals, and 80 mg/kg/day for the offspring.
Genotoxicity of lithium bromide was studied by a reverse mutation test in bacteria and by a chromosomal aberration test in cultured Chinese hamster lung (CHL/IU) cells.
Lithium bromide was not mutagenic in Salmonella typhimurium TA100, TA1535, TA98, TA1537 and Escherichia coli WP2 uvrA, with or without an exogenous metabolic activation system.
Lithium bromide did not induce chromosomal aberrations in CHL/IU cells, with or without an exogenous metabolic activation system.
| Purity | : | 55.6 % |
| Test species/strain | : | Rat/Crj:CD(SD) |
| Test method | : | OECD Test Guideline 401 |
| ””Route | : | Oral (gavage) |
| ””Dosage | : | 0 (vehicle), 670, 930, 1300, 1800, 2500 mg/kg |
| ””Number of animals/group | : | Males, 5; females, 5 |
| ””Vehicle | : | Water for injection |
| GLP | : | Yes |
””Test results:
Dead animals showed a decrease in locomotor activity, sedation and passivity. In addition, some of them demonstrated cyanosis, hypothermia and incomplete eyelid opening. From these findings, causes of death in these animals were suspected to be reduction in viability by inhibition of the central nervous system functions and disturbance of feeding behavior. Since some animals showed large amounts of nasal discharge and lacrimation before death, an abnormality in the body fluid balance could be added on a cause of death. There were no abnormal necropsy findings in dead animals, except for pale or small spleens in the males given 1300 mg/kg.
Most males and all females given 930 mg/kg or more showed decrease in locomotor activity after administration of lithium bromide, and some of them showed stereotyped behavior of rubbing the cage floor with their chins. With time, some animals became sedated, and some of them showed incomplete eyelid opening or an abnormal gait. These findings indicated that lithium bromide targets the central nerve system. Since most treated animals showed loose or mucous stool and diarrhea, and some demonstrated decrease of fecal volume several days after the treatment, the digestive system is also a target of lithium bromide toxicity. In addition, reddish tears, piloerection and salivation were found. These clinical signs disappeared by day 5 of observation, except for abdominal distention found until necropsy in surviving females given 2500 mg/kg.
Body weight gain of both sexes of animals was suppressed in the groups given 930 mg/kg and more. However, that in the surviving females given 2500 mg/kg increased after withdrawal of insult.
No abnormalities were found on necropsy of the surviving animals.
| Purity | : | 55.6 % |
| Test species/strain | : | Rat/Crj:CD(SD) |
| Test method | : | OECD Test Guideline 422 |
| ””Route | : | Oral (gavage) |
| ””Dosage | : | 0 (vehicle), 5, 20, 80 mg/kg |
| ””Number of animals/group | Males, 13; females, 13 | |
| ””Administration period | : | Males, 42 days Females, from 14 days before mating to day 4 of lactation |
| ””Terminal killing | : | Males, day 43 Females, day 5 of lactation Offspring, 4 days after birth |
| ””Vehicle | : | Water for injection |
| GLP | : | Yes |
””Test results:
In males, neither deaths nor a moribund condition were observed. Stereotyped behavior was observed in all the animals in the 80 mg/kg-treated group from the latter period of treatment onward. Body weight gain and food consumption were increased in the 80 mg/kg-treated group in the early period of treatment, and cumulative body weight gain was higher than in the controls until just before necropsy. Results of urinalysis on day 38 were normal, except that urinary specific gravity was decreased in the 20 mg/kg and more-treated groups. In the 80 mg/kg-treated group, urinary volume and potassium excretion for 24 hours were increased as well.
No effects of lithium bromide-treatment were observed on hematological parameters including coagulating time in any group, except for an increase in the percentage of monocytes in the 80 mg/kg-treated group. As for the blood biochemistry, a slight increase in creatinine concentration was observed in the 20 mg/kg and more-treated groups. Furthermore, an increase in the triglyceride concentration, slight reduction in the sodium concentration and reduction in the chloride concentration were observed in the 80 mg/kg-treated group. As for organ weights, reduction in renal weight relative to the body weight was observed in the 20 mg/kg and more-treated groups. In addition, decreases in absolute and relative pituitary weights and an increase in absolute liver weights were observed in the 80 mg/kg-treated group. No abnormalities caused by the treatment were found on pathological examination.
In females, neither deaths nor a moribund condition were observed. Stereotyped behavior was observed in most of the animals in the 80 mg/kg-treated group during the middle period of the treatment. One animal in the 80 mg/kg-treated group exhibited an impaired general condition after delivery, and showed reddish tears, emaciation, decrease in locomotor activity, sedation and cessation of fecal excretion until the day of the scheduled necropsy. This case was diagnosed as azotemia from the blood-biochemistry analysis. However, no relation to lithium bromide-treatment was suggested.
In the 80 mg/kg-treated group, body weight gain and food consumption were increased during the pre-mating period. Neither the quality nor the specific gravity of fresh urine collected on day 31 were affected by the treatment. No effects of the lithium bromide-treatment were observed on the hematological parameters at the scheduled necropsy on day 5 of lactation including coagulating time. As for the blood biochemistry, a slight reduction in the albumin concentration was observed in the 20 mg/kg and more-treated groups. Furthermore, slight reduction in concentrations of blood urea nitrogen and creatinine and a reduction in the chloride concentration were observed in the 80 mg/kg-treated group. However, there were no significant differences in organ weights between the control and lithium bromide-treated groups. No abnormal findings caused by the treatment were found on pathological examination.
<Reproductive and developmental toxicity>
No abnormalities were found regarding the estrous cycle. Some animals in the 20 mg/kg and more-treated groups did not copulate on the first estrous day during the mating period, and the numbers of vaginal estrus and pairing days until copulation tended to be increased in these groups. However, statistical differences were not found in these parameters from those in the control group. Indices of copulation index and fertility were not affected by the lithium bromide-treatment.
The treatment did not cause abnormal delivery conditions, and gestation length and gestation index were comparable to those in the controls. In addition, no effects of the treatment were observed on parameters indicating viability of embryos/fetuses until delivery, such as numbers of corpora lutea and implantations, the implantation index, number of newborn and the delivery index, and on those indicating postnatal viability, such as number of live newborn, indices of birth and live birth, number of live pups on day 4 of lactation, sex ratios on days 0 and 4 of lactation and viability index on day 4 of lactation.
No effects of the treatment on pup body weights and their morphology were observed.
<Evaluation>
From the above results, it is considered that the no observed effect dose level (NOEL) for repeat dose toxicity of lithium bromide is 5 mg/kg/day in both sexes of animals. The reasons to determine for this conclusion for males were reduction in urine specific gravity, changes in urinal and plasma creatinine concentrations and reduction in relative renal weight to the body weight in the 20 mg/kg and more-treated groups. In females, reduction in plasma concentrations of albumin were evident in these groups. The NOEL for reproductive/developmental toxicity is considered to be 5 mg/kg/day in both sexes of animals, because the pairing days until copulation tended to be prolonged in the 20 mg/kg and more-treated groups. The NOEL for the offspring is considered to be 80 mg/kg/day.
| Purity | : | 55.6 % |
| Test species/strain | : | Salmonella typhimurium TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 471 |
| ””Procedures | : | Pre-incubation method |
| ””Vehicle | : | Distilled water |
| ””Positive controls | : | -S9 mix; 2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide (TA100, TA98, WP2 uvrA), Sodium azide (TA1535) and 9-Aminoacridine (TA1537) +S9 mix; 2-Aminoanthracene (all strains) |
| ””Dosage | : | -S9 mix; 0, 313, 625, 1250, 2500, 5000 ¦Ģg/plate +S9 mix; 0, 313, 625, 1250, 2500, 5000 ¦Ģg/plate |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
””Test results:
Genotoxic effects :
S. typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
E. coli WP2 uvrA
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 55.6 % |
| Type of cell used | : | Chinese hamster lung (CHL) cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 473 |
| ””Vehicle | : | Physiological saline |
| ””Positive controls | : | -S9 mix; 1-Methyl-3-nitro-1-nitrosoguanidine +S9 mix; 3,4-Benzo[a]pyrene |
| ””Dosage | : | -S9 mix (6 hr short-term treatment); 0, 217.5, 435, 870 ¦Ģg/mL +S9 mix (6 hr short-term treatment); 0, 217.5, 435, 870 ¦Ģg/mL -S9 mix (24 hr continuous treatment); 0, 217.5, 435, 870 ¦Ģg/mL |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
””Test results:
Cytotoxicity was not observed after 6 hr short-term treatment, with or without S9 mix, and 24 hr continuous treatment without S9 mix.
Genotoxic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| 1) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257-8523, Japan. Tel +81-463-82-4751 Fax +81-463-82-9627 |
| 2) | The tests were performed by the Research Institute for Animal Science in Biochemistry and Toxicology, 3-7-11 Hashimotodai, Sagamihara-shi, Kanagawa 229-1132, Japan. Tel +81-42-762-2775 Fax +81-42-762-7979 |