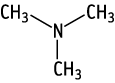

A combined repeat dose and reproductive/developmental toxicity screening test was performed with orally administered trimethylamine at doses of 0, 8, 40, 200 mg/kg. Two males and one female died in the 200 mg/kg group. Stridor and temporary salivation, decrease in total protein and albumin concentrations, inflammation, ulceration and/or hyperplasia of squamous epithelium and edema of submucosa in the gastrointestinal tract were observed in the 200 mg/kg group. The no observed effect dose level (NOEL) for systemic toxicity of trimethylamine is considered to be 40 mg/kg/day. NOELs for reproductive and developmental toxicity are considered to be 200 mg/kg/day in males and females, and 200 mg/kg/day in pups, respectively.
Reverse mutation assays using microorganisms (Salmonella typhimurium, Escherichia coli) were conducted to assess the potential of trimethylamine to induce gene mutations. Trimethylamine did not induce gene mutations in bacteria under the conditions of this study.
In vitro chromosomal aberration tests using cultured cells (CHL/IU) were conducted to assess the potential of trimethylamine to induce chromosomal aberrations. Trimethylamine induced chromosomal aberrations in cultured cells under the conditions of this study.
| Purity | : | 30.8 %(water solution) |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 401 |
| ¡¡Route | : | Oral(gavage) |
| ¡¡Doses | : | 125, 200, 320, 512, 820, 1310 mg/kg |
| ¡¡Number of animals/group | : | Males, 5; females, 5 |
| ¡¡Vehicle | : | Water for injection |
| GLP | : | Yes |
¡¡Test results:
The LD50 values were 396.9 mg/kg (95 % confidence limit:271.0¡Á580.2 mg/kg) for males and between 512 to 820 mg/kg for females.
| Purity | : | 30.8 %(water solution) |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 422 |
| ¡¡Route | : | Oral(gavage) |
| ¡¡Doses | : | 0(control), 8, 40, 200 mg/kg/day |
| ¡¡Number of animals/group | : | Males, 13; females, 13 |
| ¡¡Vehicle | : | Water for injection |
| ¡¡Administration period | : | Males, 42 days Females, from 14 days before mating to day 4 of lactation |
| GLP | : | Yes |
¡¡Test results:
The compound exerted no adverse effects on the estrous cycle, copulation and fertility, maintenance of pregnancy, parturition and lactation, as well as number of pups, delivery index, duration of pregnancy, number of corpora lutea and the implant rate at any dose level. The compound did not demonstrate any adverse effects on birth index, number of live pups, viability, sex ratio, body weight and morphological appearance of pups.
The no observed effect dose level (NOEL) for systemic toxicity of trimethylamine is considered to be 40 mg/kg/day in males and females. NOELs for reproductive and developmental toxicity are considered to be 200 mg/kg/day in males and females, and 200 mg/kg/day in pups, respectively.
| Purity | : | 30.8 % |
| Test species/strains | : | Salmonella typhimurium TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals(Chemical Substances Control Law of Japan) and OECD Test Guideline 471 |
| ¡¡Procedures | : | Pre-incubation method |
| ¡¡Solvent | : | Water for injection |
| ¡¡Positive controls | : | -S9 mix; 2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide (TA100, TA98 and WP2 uvrA), Sodium azide (TA1535) and
9-Aminoacridine hydrochloride (TA1537) +S9 mix; 2-Aminoanthracene (all strains) |
| ¡¡Doses | : | -S9 mix; 0, 39.1, 78.1, 156, 313, 625, 1250 ¦Ìg/plate(all strains) +S9 mix; 0, 39.1, 78.1, 156, 313, 625, 1250 ¦Ìg/plate(TA100, TA1535, TA1537) +S9 mix; 0, 156, 313, 625, 1250, 2500, 5000 ¦Ìg/plate(WP2 uvrA) +S9 mix; 0, 78.1, 156, 313, 625, 1250, 2500, 5000 ¦Ìg/plate(TA98) |
| ¡¡S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ¡¡Plates/test | : | 3 |
| ¡¡Number of replicates | : | 2 |
| GLP | : | Yes |
¡¡Test results:
Genetic effects:
Salmonella typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| ¡¡Without metabolic activation: | [ ] | [ ] | [*] |
| ¡¡With metabolic activation: | [ ] | [ ] | [*] |
Escherichia coli WP2 uvrA
| + | ? | - | |
| ¡¡Without metabolic activation: | [ ] | [ ] | [*] |
| ¡¡With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 30.8 % |
| Type of cell used | : | Chinese hamster lung (CHL/IU) cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 473 |
| ¡¡Solvent | : | Physiological saline |
| ¡¡Positive controls | : | -S9 mix; Mitomycin C +S9 mix; Cyclophosphamide |
| ¡¡Doses | : | -S9 mix(short-term treatment); 0, 148, 296, 591 ¦Ìg/mL +S9 mix(short-term treatment); 0, 148, 296, 591 ¦Ìg/mL -S9 mix(short-term treatment, confirmative test); 0, 378, 473, 591 ¦Ìg/mL +S9 mix(short-term treatment, confirmative test); 0, 378, 473, 591 ¦Ìg/mL |
| ¡¡S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ¡¡Plates/test | : | 2 |
| GLP | : | Yes |
¡¡Test results:
Genetic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| ¡¡Without metabolic activation: | [*] | [ ] | [ ] | [ ] | [ ] | [*] |
| ¡¡With metabolic activation: | [*] | [ ] | [ ] | [ ] | [ ] | [*] |
| 1) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257-0025, Japan. Tel +81-463-82-4751 Fax +81-463-82-9627 |
| 2) | The tests were performed by the Biosafety Research Center, Foods, Drugs and Pesticides (An-pyo Center), 582-2 Shioshinden, Fukude-cho, Iwata-gun, Shizuoka, 437-1213, Japan. Tel +81-538-58-1266 Fax +81-538-58-1393 |