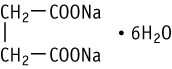
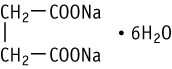
Disodium succinate hexahydrate was studied for oral toxicity in rats in an OECD combined repeated dose and reproductive/developmental toxicity screening test at doses of 0, 100, 300 and 1000 mg/kg.
With regard to repeated dose toxicity, loosening of stools was observed in males of the 1000 mg/kg group and an increase of blood urea nitrogen was evident in females of the 1000 mg/kg group. On urinalysis, a few males showed high values for protein or were positive for occult blood in the 300 and 1000 mg/kg groups.
The NOELs for repeated dose toxicity are considered to be 100 mg/kg/day for males, and 300 mg/kg/day for females.
With regard to reproductive/developmental toxicity, no adverse effects were observed in terms of the estrus cycle, copulation and fertility results or findings on delivery.
No abnormal findings related to the test substance were noted on external examination, or in terms of clinical signs, viability, growth or necropsy findings for the offspring.
The NOELs for reproductive and developmental toxicity are considered to be 1000 mg/kg/day for both parental animals and offspring.
Reverse mutation assays using microorganisms (Salmonella typhimurium TA100, TA1535, TA98, TA1537 and Escherichia coli WP2 uvrA) were conducted to assess the potential of disodium succinate hexahydrate to induce gene mutations. No mutagenic activity was found in bacteria under the present experimental conditions.
In vitro chromosomal aberration tests using cultured mammalian cells (CHL/IU) were conducted to assess the potential of disodium succinate hexahydrate to induce chromosomal aberrations. Neither structural nor numerical chromosome aberrations were detected under the present experimental conditions.
| Purity | : | 99.9 wt% |
| Test species/strain | : | Rat/Crj:CD(SD) |
| Test method | : | OECD Test Guideline 401 |
| ””Route | : | Oral (gavage) |
| ””Dosage | : | 0 (vehicle), 2000 mg/kg |
| ””Number of animals/group | : | Males, 5; females, 5 |
| ””Vehicle | : | Distilled water |
| GLP | : | Yes |
””Test results:
LD50 values were found to be higher than 2000 mg/kg for both sexes.
| Purity | : | 99.9 wt% |
| Test species/strains | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 422 |
| ””Route | : | Oral (gavage) |
| ””Dosage | : | 0 (vehicle), 100, 300, 1000 mg/kg |
| ””Number of animals/group | Males, 12; females, 12 | |
| ””Vehicle | : | Water for injection |
| ””Administration period | : | Males, 52 days Females, from 14 days before mating to day 4 of lactation |
| ””Terminal killing | : | Males, day 53 Females, day 5 of lactation |
| GLP | : | Yes |
””Test results:
No deaths were observed in any treatment group in either sex. Loosening of the stools was observed in males of the 1000 mg/kg group. The test substance had no effects on body weights, food consumption or hematological parameters in any treatment group. Increase of blood urea nitrogen was observed in females of the 1000 mg/kg group. On urinalysis, a few males showed high values for protein or were positive for occult blood in the 300 and 1000 mg/kg groups.
No effects related to the test substance were observed with regard to organ weights, necropsy or histological findings. No abnormalities were noted regarding cycling in the seminiferous epithelium.
The NOELs for repeated dose toxicity are considered to be 100 mg/kg/day for males, and 300 mg/kg/day for females.
<Reproductive and Developmental toxicity>
No adverse effects were observed in terms of the estrus cycle, copulation, fertility results, delivery conditions, gestation length, numbers of corpora lutea or implantations, the sex ratio, implantation index, gestation index, livebirth index or the delivery index.
No abnormal findings related to the test substance were noted on external examination, or in terms of clinical signs, viability, growth or necropsy findings for the offspring.
The NOELs for reproductive and developmental toxicity are considered to be 1000 mg/kg/day for both parental animals and offspring.
| Purity | : | 99.9 wt% |
| Test species/strains | : | Salmonella typhimurium TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 471 |
| ””Procedures | : | Pre-incubation method |
| ””Solvent | : | Saline |
| ””Positive controls | : | -S9 mix; 2-(2-Furyl)-3-(5-nitro-2-furyl) acrylamide (TA100, TA98 and WP2 uvrA), Sodium azide (TA1535) and 2-Methoxy-6-chloro-9-[3-(2-chloroethyl)-aminopropylamino] acridine”¦2HCl (TA1537) +S9 mix; Benzo[a]pyrene (TA100, TA98, TA1537) and 2-Aminoanthracene (TA1535 and WP2 uvrA) |
| ””Dosage | : | -S9 mix; 0, 156, 313, 625, 1250, 2500 and 5000 ¦Ģg/plate (all strains) +S9 mix; 0, 156, 313, 625, 1250, 2500 and 5000 ¦Ģg/plate (all strains) |
| ””S9 | : | Rat liver, induced with phenobarbital and 5, 6-benzoflavone |
| ””Plates/test | : | 3 |
| GLP | : | Yes |
””Test results:
Genetic effects:
Salmonella typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
Escherichia coli WP2 uvrA
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99.9 wt% |
| Type of cell used | : | Chinese hamster lung (CHL/IU) cell |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 473 |
| ””Solvent | : | Saline |
| ””Positive control | : | S9 mix; Mitomycin C +S9 mix; Cyclophosphamide |
| ””Dosage | : | -S9 mix (short-term treatment); 0, 313, 625, 1250, 2500 and 5000 ¦Ģg/mL +S9 mix (short-term treatment); 0, 313, 625, 1250, 2500 and 5000 ¦Ģg/mL -S9 mix (continuous treatment 24 and 48 hrs); 0, 313, 625, 1250, 2500 and 5000 ¦Ģg/mL |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
””Test results:
Genotoxic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| 1) | The tests were performed by the Biosafety Research Center, Foods, Drugs and Pesticides(An-pyo Center), 582-2 Shioshinden, Fukude-cho, Iwata-gun, Shizuoka, 437-1213, Japan. Tel +81-538-58-1266 Fax +81-538-58-1393 |
| 2) | The tests were performed by the Bozo Research Center Inc., 1284 Kamado, Gotemba-shi, Shizuoka, 412-0039, Japan. Tel & Fax +81-550-82-9922 |