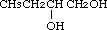
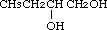
1,2-Butanediol was studied for oral toxicity in rats in an OECD combined repeat dose and reproductive/developmental toxicity screening test at doses of 40, 200 and 1000 mg/kg/day, and for mutagenicity with assays for reverse mutation in bacteria and chromosomal aberrations in cultured Chinese hamster (CHL) cells.
For repeat dose toxicity, 1,2-butanediol was found to cause only slight clinical signs including transient hypolocomotion and hypopnea among the females given 1000 mg/kg. There were no significant differences between the treated and control animals of both sexes in body weight, food consumption, hematological and blood chemical parameters, organ weight, or histopathological findings. NOELs for repeat dose toxicity were 1000 mg/kg/day in males and 200 mg/kg/day in females. With regard to reproductive/developmental toxicity, 1,2-butanediol showed no effects on reproductive parameters (copulation, implantation, pregnancy, parturition, or lactation) or on offspring. NOELs for reproductive performance and offspring development were both 1000 mg/kg/day.
1,2-Butanediol was not mutagenic to S. typhimurium TA100, TA98, TA97 and TA102 with or without exogenous metabolic activation up to 5000 ¶Őg/plate. This chemical induced neither chromosomal aberrations nor polyploidy in CHL cells with or without exogenous metabolic activation up to 0.9 mg/ml.
| Purity | : | > 99 % |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | OECD Combined Repeat Dose and Reproductive/Developmental Toxicity Screening Test |
| °°Route | : | Oral (gavage) |
| °°Doses | : | 0 (vehicle), 40, 200, 1000 mg/kg/day |
| °°Number of animals | : | Male;10, Female10 |
| °°Vehicle | : | Distilled water |
| °°Administration period | : | Male, 42 days Female, from 14 days before mating to day 3 of lactation |
| °°Terminal kill | : | Male, day 43 Female, day 4 of lactation |
| GLP | : | Yes |
°°Test results:
| Purity | : | > 99% |
| Test species/strains | : | S. typhimurium TA100, TA1535, TA98, TA1537 E. coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| °°Procedure | : | Plate method |
| °°Solvent | : | DMSO |
| °°Positive controls | : | -S9, AF-2 (TA100,WP2,TA98), sodium azide (TA1535) and 9-aminoacridine (TA1537) +S9, 2-Aminoanthracene (all strains) |
| °°Doses | : | 0, 312.5, 625, 1250, 2500, 5000 ¶Őg/plate |
| °°S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| °°Plates/test | : | 3 |
| °°Number of replicates | : | 2 |
| GLP | : | Yes |
Genotoxic effects:
S. typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| with metabolic activation: | [ ] | [ ] | [*] |
| without metabolic activation: | [ ] | [ ] | [*] |
E. coli WP2 uvrA
| + | ? | - | |
| with metabolic activation: | [ ] | [ ] | [*] |
| without metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | >99 % |
| Type of cell used | : | Chinese hamster CHL cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| °°Solvent | : | DMSO |
| °°Positive controls | : | -S9, Mitomycin C +S9, Cyclophosphamide |
| °°Doses | : | -S9: 0, 0.23, 0.45, 0.90 mg/ml +S9: 0, 0.23, 0.45,0.90 mg/ml |
| °°S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| °°Plates/test | : | 2 |
| GLP | : | Yes |
| + | ? | - | |
| with metabolic activation: | [ ] | [ ] | [*] |
| without metabolic activation: | [ ] | [ ] | [*] |
| 1) | The test was performed by the Mitsubishi-Kasei Institute of Toxicological and Environmental Sciences, 14 Sunayama, Hazaki-machi, Kashima-gun, Ibaraki, 314-02, Japan. Tel 81-479-46-2871 Fax 81-479-46-2874 |
| 2) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257, Japan. Tel 81-463-82-4751 Fax 81-463-82-9627 |