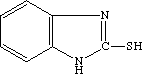
2-Mercaptobenzimidazole
2-„į„ė„«„ׄȄŁ„ó„Ä„¤„ß„Ą„¾”¼„ė
CAS No. 583-39-1
2-Benzimidazolethiol
2-„Ł„ó„Ä„¤„ß„Ą„¾”¼„ė„Į„Ŗ”¼„ė
Molecular formula: C7H6N2S””””Molecular weight: 150.21

The single dose oral toxicity test revealed LD50 values of 218 mg/kg for males and 208 mg/kg for females.
With regard to repeat dose toxicity, one female receiving 40 mg/kg died on Day 24 of administration. Suppression of body weight gain and reduced food consumption were noted in males given 12 mg/kg and in both sexes given 40 mg/kg. Decreased values for PLT, RET, WBC and RBC counts, HGB, MCV, and elevated values for MCHC, PT and APTT were obtained with males and/or females at treated with 12 mg/kg or more. Lower than normal values for K, Ca, GOT, Cl, IP, a2-globulin ratio, b-globulin ratio, and increased g-GTP, TP, ALB, albumin, the A/G ratio, T-BIL, BUN, CRE, GLU, T-CHO, and Na were obtained with males and/or females receiving 4 mg/kg or more. Enlargement of the thyroid was found in both sexes at doses of 4 mg/kg and above, and higher values for absolute and relative thyroid weights were obtained for males given 4 mg/kg and both sexes receiving 12 mg/kg or more. Both sexes dosed with 1.2 mg/kg or more showed hyperplasia/hypertrophy of follicular cells in the thyroids. NOELs for repeat dose toxicity are considered to be less than 1.2 mg/kg in both sexes.
2-Mercaptobenzimidazole was not mutagenic in Salmonella typhimurium TA100, TA98, TA1535, TA1537 and Escherichia coli WP2 uvrA.
This chemical substance induced structural chromosomal aberrations in CHL/IU cells in all groups (0.4-1.5 mg/ml) treated for 6-hr with exogenous metabolic activation, but not in the absence of an exogenous metabolic activation system. Polyploid cells were also marginally induced in all groups with a metabolic activation system, the frequencies ranging from 4.0 to 4.75%.
| Purity | : | 98.5% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | OECD Test Guideline 401 |
| ””Route | : | Oral (gavage) |
| ””Dosage | : | 0 (Vehicle), 79, 119, 178, 267, 400, 600 mg/kg |
| ””Number of animals | : | Males,5;Females,5/group |
| ””Vehicle | : | 0.5% CMC |
| GLP | : | Yes |
LD50 values were found to be 218 (166-287) mg/kg for the males and 208 mg/kg for the females.
| Purity | : | 98.5% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | Guideline for the 28-Day Repeat Dose Toxicity Test of Chemicals (Japan) |
| ””Route | : | Oral (gavage) |
| ””Doses | : | 0 (Vehicle), 1.2, 4, 12, 40 mg/kg/day |
| ””Number of animals | : | Males, 10 or 15 (0, 40 mg/kg) Females, 10 or 15 (0, 40 mg/kg) |
| ””Vehicle | : | 0.5% CMC |
| ””Administration period | : | Males, Females; 28 days |
| ””Terminal kill | : | Males, Females; days 29 or 43 (0, 40 mg/kg) |
| GLP | : | Yes |
Suppression of body weight gain was found in males given 12 mg/kg from Day 18, in males given 40 mg/kg from Day 11 and in females given 40 mg/kg from Day 15 of the administration. Lower than normal body weights were continuously obtained for both sexes receiving 40 mg/kg during the recovery period.
Lower food consumption was noted for males given 12 mg/kg from Week 2 and for both sexes given 40 mg/kg from Week 1. Lower food consumption values were also continuously obtained for both sexes given 40 mg/kg during the recovery period.
Increased urine volume was noted in males given 40 mg/kg, and lower values for urinary specific gravity in males receiving 12 mg/kg or more. Both retuned to normal during the recovery period.
On hematological examination, the following changes were found : lower values for platelet and reticulocyte counts and higher MCHC values in males given 12 mg/ kg or more and in females given 40 mg/kg, lower MCV values in males given 12 mg/kg or more, lower RBC counts in females given 12 mg/kg or more, lower values for HCT and prolonged PT in both sexes receiving 40 mg/kg, prolonged APTT in males given 40 mg/kg, and lower WBC counts in females given 40 mg/kg. After the recovery period, lower HCT values and RBC counts were also found in both sexes given 40 mg/kg and in the high dose, respectively, and lower RBC, HGB and WBC count receiving were newly found in males, both sexes and males, respectively, 40 mg/kg.
On blood chemical analysis, the following changes were found : lowered values of K in both sexes given 4 mg/kg or more, lowered Ca in males given 4 mg/kg or more, lowered Cl and GOT in both sexes given 12 mg/kg or more, higher TP, BUN, CRE and T-CHO in males receiving 12 mg/kg or more and in the 40 mg/kg females, higher values for ¦Ć-GTP and ALB in both sexes given 40 mg/kg, lower values for ¦Į2-globulin, and ¦Ā-globulin TG and IP in males given 12 mg/kg or more, higher values for albumin, the A/G ratio and T-Bil in males receiving 40 mg/kg, higher GLU in females given 40 mg/kg and elevated Na in females receiving 12 mg/kg or more. After the recovery period, decreased GOT and b-globulin were also found in males receiving 40 mg/kg, and increased Na was newly found in males of the same group.
Enlargement of thyroids was found in both sexes receiving 4 mg/kg or more at necropsy, and also after the recovery period.
Higher values for absolute and relative thyroid weights were found in males receiving 4 mg/kg or more and in females given 12 mg/kg or more. After the recovery period, elevated values for both parameters were still found, albeit to a lesser extent, in both sexes given 40 mg/kg.
On histopathological examination, both sexes receiving 1.2 mg/kg or more showed hyperplasia/hypertrophy of follicular cells in the thyroids. Animals receiving 40 mg/kg showed vacuolization of cortical cells in the adrenal glands. The histopathological changes in the thyroids and adrenal glands were still present although weaker in degree after the recovery period.
Based on the above results, we conclude that 2-mercaptobenzimidazole affects the thyroids, hematopoietic functions, and hepatic functions, renal functions. NOELs of 2-mercaptobenzimidazole for 28-day repeat dose oral administration are considered to be less than 1.2 mg/kg for both sexes under the conditions of the present study.
| Purity | : | 98.5% |
| Test species/strains | : | S.typhimurium TA100, TA1535, TA98, TA1537 E. coli WP2 uvrA |
| Test methods | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ””Procedures | : | Plate incorporation method |
| ””Solvent | : | DMSO |
| ””Positive controls | : | -S9 Mix, AF-2 (TA100, WP2, TA98) sodium azide (TA1535) and 9-aminoacridine (TA1537) +S9 Mix, 2-aminoanthracene (all strains) |
| ””Doses | : | 0, 312.5, 625, 1250, 2500, 5000 ¦Ģg/plate |
| ””S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
| + | ? | - | |
| without metabolic activation: | [ ] | [ ] | [*] |
| with metabolic activation: | [ ] | [ ] | [*] |
E. coli WP2 uvrA
| without metabolic activation: | [ ] | [ ] | [*] |
| with metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 98.5% |
| Type of cell used | : | Chinese hamster lung (CHL/IU) cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ””Solvent | : | Dimethyl sulfoxide |
| ””Positive controls | : | -S9, Mitomycin C +S9, Cyclophosphamide |
| ””Doses | : | -S9 (continuous treatment): 0, 0.10, 0.20, 0.39 mg/ml -S9 (short-term treatment): 0, 0.4, 0.8, 1.5 mg/ml +S9 (short-term treatment): 0, 0.4, 0.8, 1.5 mg/ml |
| ””S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
| without metabolic activation (continuous treatment ): | 0.39 mg/ml (clastogenicity) |
| with metabolic activation (short-term treatment): | 0.8 mg/ml (clastogenicity) 0.4 mg/ml (polyploidy) |
Genotoxic effects:
| clastogenicity | ””polyploidy”” | ||||||
| + | ? | - | ”” | + | ? | - | |
| without metabolic activation: | [ ] | [*] | [ ] | ”” | [ ] | [ ] | [*] |
| with metabolic activation: | [*] | [ ] | [ ] | ”” | [ ] | [*] | [ ] |
| 1) | The test was performed by Nihon Bioresearch Inc. Hashima Laboratory, 6-104 Majima, Fukuju-cho, Hashima, Gifu, 501-62 Japan. Tel +81-58-392-6222 Fax +81-58-392-1284 |
| 2) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257 Japan, Tel +81-463-82-4751 Fax +81-463-82-9627 |