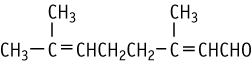
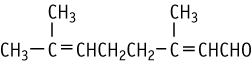
Males and females given 1000 mg/kg/day showed decreases in body weights and food consumption. Autopsy revealed partial thickening of mucosal layer in the stomach in females given 1000 mg/kg, and histopathological examination of the forestomach demonstrated squamous hyperplasia, ulceration and granulation in the lamina propria in males and/or females given 1000 mg/kg.
The NOEL for repeated dose toxicity is considered to be 200 mg/kg/day for both sexes.
No adverse effects were detected in terms of ability, organ weights or histopathology of the reproductive organs for parental animals, and of delivery or maternal behavior for dams. Male and female pups showed lowering of body weights in the 1000 mg/kg group, but no other adverse effects were detected.
The NOELs for reproductive/developmental toxicity are considered to be 1000 mg/kg/day for both parental animals, and 200 mg/kg/day for offspring.
| Purity | : | 98.20 % |
| Test species/strain | : | Rat/Crj:CD(SD) |
| Test method | : | OECD Test Guideline 421 |
| ¡¡Route | : | Oral (gavage) |
| ¡¡Dosage | : | 0 (vehicle), 40, 200, 1000 mg/kg |
| ¡¡Number of animals/group | : | Males, 12; females, 12 |
| ¡¡Vehicle | : | Corn oil |
| ¡¡Administration period | : | Males, 46 days Females, from 14 days before mating to day 3 of lactation |
| ¡¡Terminal killing | : | Males, day 47 Female, day 4 of lactation |
| GLP | : | Yes |
¡¡Test results:
Males and females given 1000 mg/kg showed decreases in body weights and food consumption. Autopsy revealed partial thickening of mucosal layer in the stomach in females given 1000 mg/kg, and histopathological examination of the forestomach demonstrated squamous hyperplasia, ulceration and granulation in the lamina propria in males and/or females given 1000 mg/kg.
On the basis of these findings, the NOEL for repeated dose toxicity is considered to be 200 mg/kg/day for both sexes.
<Reproductive and developmental toxicity>
No adverse effects were detected in terms of reproductive ability, organ weights or histopathology of the reproductive organs for parental animals, and of delivery or maternal behavior for dams. Male and female pups showed lowering of body weights in the 1000 mg/kg group, but no other adverse effects were detected.
On the basis of these findings, the NOELs for reproductive/developmental toxicity are considered to be 1000 mg/kg/day for both parental animals, and 200 mg/kg/day for offspring.
| 1) | The test was performed by the Safety Research Institute for Chemical Compounds Co., Ltd., 363-24 Shin-ei, Kiyota-ku, Sapporo, Hokkaido, 004-0839, Japan. Tel +81-11-885-5031 Fax +81-11-885-5313 |