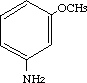

3-Methoxybenzenamine was studied for oral toxicity in rats in a single dose toxicity study at doses of 200, 300, 500, 800,1300 and 2000 mg/kg and in an OECD combined repeat dose and reproductive/developmental toxicity screening test at doses of 0, 2.4, 12, 60 and 300 mg/kg/day. 3-Methoxybenzenamine was also tested for mutagenicity with assays for reverse mutation in bacteria, chromosomal aberrations in cultured Chinese hamster (CHL) cells and induction of micronuclei in mice.
The single dose oral toxicity revealed the LD50s of 526 mg/kg when administerd in corn oil and above 300 mg/kg in both sexes with distilled water as the vehicle.
For repeat dose toxicity, 3-methoxybenzenamine caused changes such as salivation, brown urine, hemorrhage from the vagina, lowering of body weight gain, decrease in food consumption and splenomegaly in the 300 mg/kg/day males and females. The males at doses of 60 mg/kg or more showed hemolytic anemia, significant increases in blood total bilirubin, inorganic phosphorus and Na, decreases in total protein, glucose and Ca, and congestion and decreased cellularity of B-cell areas evident on histopathological examination of spleens. Extramedullary hematopoiesis and brown pigmentation were observed in the 2.4 mg/kg or higher and the 12 mg/kg or higher dose groups, respectively. These findings suggest that 3-Methoxybenzenamine affects the hematopoietic system, and hepatic and renal functions. NOEL for repeat dose toxicity was less than 2.4 mg/kg/day in males and females. For reproductive/developmental toxicity, all dams in the 300 mg/kg group failed to deliver pups. Their copulation and fertility, however, were not affected. Since necropsy of these dams revealed neonatal death in the early pregnancy period, the test chemical is suggested to have lethal effects on the fetus or neonates. NOELs for reproductive performance were 300 mg/kg/day in males and 60 mg/kg/day in females. The NOEL for offspring development was 60mg/kg/day.
3-Methoxybenzenamine was mutagenic for S. typhimurium, TA98 with metabolic activation, and not mutagenic for TA100, TA97 and TA102 with or without exogenous metabolic activation up to 5000 ¦Ģg/plate. This chemical induced chromosomal aberrations in CHL cells without exogenous metabolic activation. Polyploidy was not evident under the assay conditions. The test chemical also induced micronucleated polychromatic erythrocytes in male BDF1 mouse bone marrow 72 h after treatment at a dose of 800 mg/kg.
| Purity | : | > 98% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | OECD Guideline 401 |
| ””Doses | : | 200, 300, 500, 800, 1300, 2000 mg/kg (in corn oil) 200, 300 mg/kg (in distilled water) |
| ””Number of animals | : | Male, 5; Female, 5/group |
| GLP | : | Yes |
| Test results | : | LD50: LD50: Male and Female, 526 mg/kg in corn oil Male and Female, 300 mg/kg or more in distilled water |
| Purity | : | > 98 % |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | OECD Combined Repeat Dose and Reproductive/Developmental Toxicity Screening Test |
| ””Route | : | Oral (gavage) |
| ””Doses | : | 0 (vehicle), 2.4, 12, 60, 300 mg/kg/day |
| ””Number of animals | : | Male,12; Female,12 |
| ””Vehicle | : | Corn oil |
| ””Administration period | : | Male, 50 days Female, from 14 days before mating to day 3 of lactation |
| ””Terminal kill | : | Male, day 51 Female, day 4 of lactation |
| GLP | : | Yes |
””Test results:
| Purity | : | 99 % |
| Test species/strains | : | S. typhimurium TA100, TA98, TA102, TA97 |
| Test method | : | Maron & Ames (1983) |
| ””Procedure | : | Preincubation assay |
| ””Solvent | : | DMSO |
| ””Positive controls | : | -S9, AF-2 (TA100, TA98) ICR-191 (TA97) Mitomycin C (TA102) +S9, 2-Aminoanthracene (all strains) |
| ””Doses | : | 0, 25, 50, 100, 250, 500, 1000, 2500, 5000 ¦Ģg/plate |
| ””S-9 | : | Mouse liver, induced with KC-400 (equivalent to PCB) |
| ””Plates/test | : | 2 |
| ””Number of replicates | : | Tests for TA98 with and without metabolic activation were repeated twice. Others were performed once. |
Genotoxic effects:
TA100, TA102, TA97
| + | ? | - | |
| with metabolic activation: | [ ] | [ ] | [*] |
| without metabolic activation: | [ ] | [ ] | [*] |
TA 98
| + | ? | - | |
| with metabolic activation: | [*] | [ ] | [ ] |
| without metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99 % |
| Type of cell used | : | Chinese hamster CHL cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ””Solvent | : | DMSO |
| ””Doses | : | -S9: 0, 0.25, 0.5, 0.75 mg/ml +S9:0, 0.0313, 0.0625, 0.125 mg/ml |
| ””S-9 | : | Mouse, induced with PCB |
| ””Plates/test | : | 1 |
| + | ? | - | |
| with metabolic activation: | [ ] | [ ] | [*] |
| without metabolic activation: | [*] | [ ] | [ ] |
| Purity | : | > 98 % |
| Test species/strain | : | Mouse/Crj:BDF1 |
| Test method | : | Guidelines for Mutagenicity Testing of Chemicals (Japan) |
| ””Doses | : | Male: 0, 200, 400, 800 mg/kg Female: 0, 150, 300, 600 mg/kg |
| ””Solvent | : | Olive oil |
| GLP | : | Yes |
| + | ? | - | |
| Micronucleus test: | [*] | [ ] | [ ] |
| 1) | The tests were performed by Nihon Bioresearch Inc., Hashima Laboratory, 6-104 Majima, Fukuju-cho, Hashima, Gifu, 501-62, Japan. Tel 81-583-92-6222 Fax 81-583-92-1284 |
| 2) | The tests were performed by the Biological Safety Research Center, National Institute of Health Sciences, 1-18-1 Kamiyoga, Setagaya-ku, Tokyo, 158, Japan. Tel 81-3-3700-1141 Fax 81-3-3700-2348 |
| 3) | The test was performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257, Japan. Tel 81-463-82-4751 Fax 81-463-82-9627 |