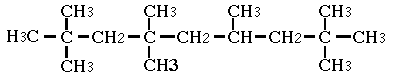
2,2,4,4,6,8,8-Heptamethylnonane was studied for oral toxicity in rats in a single dose toxicity test at doses of 500, 1000 and 2000 mg/kg and in a 28-day repeat dose toxicity test at doses of 0, 100, 300 and 1000 mg/kg/day. 2,2,4,4,6,8,8-Heptamethylnonane was also tested for mutagenicity with assays for reverse mutation in bacteria and chromosomal aberrations in cultured Chinese hamster (CHL) cells.
The single dose oral toxicity test resulted in LD50s of above 2000 mg/kg for both sexes.
In the repeat dose toxicity test, hematological analysis revealed prolongation of prothrombin time and activated partial thromboplastin times in 1000 mg/kg males at the end of the 28-day administration period and even after the 14-day recovery period. Blood chemical analyses showed decreases in triglycerides, K and Cl, and an increase in GPT in the 300 and or 1000 mg/kg groups. Absolute and relative liver weights increased in males and females of the 300 mg/kg group, and the relative weights still remained high after the recovery period in males of the 1000 mg/kg group. On histopathological examination, centrilobular hypertrophy of hepatocytes was observed in both sexes receiving 1000 mg/kg and in females given 300 mg/kg. One case showed necrosis. Scaring with pigmentation of hepatocytes were noted in one 1000 mg/kg male after the recovery period. The NOEL for repeat dose toxicity was 100 mg/kg/day.
2,2,4,4,6,8,8-Heptamethylnonane was not mutagenic for S. typhimurium TA100, TA1535, TA98, TA1537, and E. coil WP2 uvrA with or without exogenous metabolic activation up to 5000 ¦Ģg/plate. This chemical induced neither chromosomal aberrations nor polyploidy with or without exogenous metabolic activation up to the cytotoxic concentration of 2.26 mg/ml.
| Purity | : | 99.9% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | OECD Test Guideline 401 |
| ””Dose | : | 500, 1000, 2000 mg/kg |
| ””Number of animals | : | Male, 5; Female, 5/group |
| GLP | : | Yes |
| Test results | : | LD50: Male > 2,000 mg/kg; Female > 2,000 mg/kg |
| Purity | : | 99.9 % |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | Guidelines for 28-Day Repeat Dose Toxicity Test of Chemicals (Japan) |
| ””Route | : | Oral (gavage) |
| ””Doses | : | 0 (vehicle), 100, 300, 1000 mg/kg/day |
| ””Number of animals | : | Male, 5; Female, 5 |
| ””Vehicle | : | Corn Oil |
| ””Administration period | : | Male and Female, 28 days |
| ””Terminal kill | : | Male and Female, days 29 or 43 |
| GLP | : | Yes |
””Test results:
On urinalysis, a positive bilirubin reaction occurred without dose-dependency at the end of administration period, and occult blood was observed in 1000 mg/kg males after the recovery period. One of these latter cases was also found positive for glucose. Prothrombin and activated thromboplastin times were prolonged at the end of the administration period. These effects remained albeit somewhat reduced after the recovery period. On blood chemical analysis after the administration period, triglycerides were significantly decreased in 300 mg/kg and 1000 mg/kg males, GPT was significantly increased in 1000 mg/kg females, and K and Cl decreased in 1000 mg/kg males. The analyses performed after the recovery period revealed A/G ratios to be significantly lowered in 1000 mg/kg females, and LDH activity to be significantly elevated in 1000 mg/kg males.
Absolute and relative liver weights were increased in 300 mg/kg males and females after the administration period, and the relative weights remained increased in 1000 mg/kg males even after the recovery period. Histopathological findings after the administration period included, centrilobular hypertrophy of hepatocytes in 1000 mg/kg males and females and in 300mg/kg males. One case showed necrosis. In the findings after the recovery period, scarring with pigmentation of hepatocytes were noted in one of the 1000 mg/kg males. The hepatocyte hypertrophy observed after the administration had vanished.
| Purity | : | 99.9% |
| Test species/strains | : | S. typhimurium TA100, TA1535, TA98, TA1537 E. coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ””Procedure | : | Plate method |
| ””Solvent | : | Acetone |
| ””Positive controls | : | -S9, AF-2 (TA100, WP2 uvrA,TA98), sodium azide (TA1535) and 9-aminoacridine (TA1537) +S9, 2-Aminoanthracene (all strains) |
| ””Doses | : | 0, 312.5, 625, 1250, 2500, 5000 ¦Ģg/plate |
| ””S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
””Test results:
Genotoxic effects:
S. typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| with metabolic activation: | [ ] | [ ] | [*] |
| without metabolic activation: | [ ] | [ ] | [*] |
E. coli WP2 uvrA
| + | ? | - | |
| with metabolic activation: | [ ] | [ ] | [*] |
| without metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99.9% |
| Type of cell used | : | Chinese hamster CHL cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ””Solvent | : | Acetone |
| ””Positive controls | : | -S9, Mitomycin C +S9, Cyclophosphamide |
| ””Doses | : | -S9: 0, 0.57, 1.13, 2.26 mg/ml +S9: 0, 0.57, 1.13, 2.26 mg/ml |
| ””S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
””Test results:
Lowest concentration producing cytogenetic effects in vitro:
with metabolic activation: > 2.26 mg/ml
without metabolic activation: > 2.26 mg/ml
Genotoxic effects:
| + | ? | - | |
| with metabolic activation: | [ ] | [ ] | [*] |
| without metabolic activation: | [ ] | [ ] | [*] |
| 1) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257, Japan. Tel 81-463-82-4751 Fax 81-463-82-9627 |