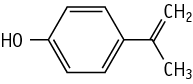

With regard to the single dose toxicity, deaths occurred in both sexes of the 600 and 1200 mg/kg groups. Clinical signs of bradypnea, hypothermia, adoption of a lateral position, decrease in locomotor activity, and soiling of perigenital fur, were observed in males and females given 600 and 1200 mg/kg from 1 to 6 hours after administration. Decrease of body weight gain was observed in surviving males and females given 600 mg/kg. Autopsy revealed dark red foci in the glandular stomach or forestomach in dead females.
The LD50 value was estimated to be 585.8 mg/kg for both sexes.
With regard to repeated dose toxicity, soiling of perigenital fur appeared in males and females at 300 mg/kg and above. Salivation also appeared in females at 300 mg/kg and above. Suppression of body weight gain and decrease of food consumption were apparent in females at 300 mg/kg and above after day 7 of the administration period.
On urinalysis, increase of urine volume was observed in males given 300 mg/kg, and males and females given 400 mg/kg. With 400 mg/kg, decrease of pH in males and females, and decrease of specific gravity in females were also observed. Water consumption was increased in males given 400 mg/kg.
On hematological examination, statistically significant decrease of MCHC and prolongation of APTT were observed in females given 400 mg/kg, while decrease of MCHC was observed also in males given 300 and 400 mg/kg after the recovery period.
On biochemical examination, increase of ¦Ć-GTP was observed in males at 300 mg/kg and above, and females at 400 mg/kg. With 400 mg/kg, increase of ¦Ā-globulin in males and of ¦Į1-globulin in females was also observed.
Thickening of mucosa in the forestomach was noted in males and females given 400 mg/kg. Dilatation and watery contents of the cecum and atrophy of the spleen and thymus were observed in one female given 400 mg/kg.
Liver weights were increased in males given 100 mg/kg, and in males and females receiving 300 or 400 mg/kg. Relative liver weight was increased in males given 300 mg/kg, and in males and females given 400 mg/kg. Absolute and relative kidney weights were increased in males given 400 mg/kg. Absolute and relative thymus weights were decreased in females given 400 mg/kg.
Histopathological examination revealed squamous cell hyperplasia of the forestomach in females at 100 mg/kg and above. Squamous cell hyperplasia at the limiting ridge of the stomach was also observed in males and females at 100 mg/kg and above. Formation of hyaline droplets and eosinophilic bodies in the renal proximal tubular epithelium was observed in males given 400 mg/kg. Atrophy of the thymus was observed in females given 400 mg/kg.
Animals demonstrated reduction or disappearance of all the charges at the end of the 14 day recovery period.
The NOEL is considered to be 30 mg/kg/day for males and females.
A preliminary reproduction toxicity screening test was conducted at doses of 0, 4, 15 and 60 mg/kg in rats following OECD test guideline 421. Transient salivation occurred after oral administration in the group receiving 60 mg/kg. Thickening of mucosa of forestomach due to squamous hyperplasia was observed in the 60 mg/kg group at autopsy. A decrease in the thymus weight was observed in females of the 60 mg/kg group. There were no adverse effects on any reproductive and developmental parameters. The no observed effect dose level (NOEL) for systemic toxicity was considered to be 15 mg/kg/day in both sexes, and that for reproductive and developmental toxicity was 60 mg/kg/day.
Reverse mutation assays using microorganisms (Salmonella typhimurium, Escherichia coli) were conducted to assess the potential of 4-(1-methylethenyl)phenol to induce gene mutations.
4-(1-Methylethenyl)phenol did not induce gene mutations in bacteria under the conditions of this study.
In vitro chromosomal aberration tests using cultured Chinese hamster cells (CHL/IU) were conducted to assess the potential of 4-(1-methylethenyl)phenol to induce chromosomal aberrations.
4-(1-Methylethenyl)phenol induced chromosomal aberrations in cultured cells under the conditions of this study.
| Purity | : | > 99 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 401 |
| ””Route | : | Oral (gavage) |
| ””Dosage | : | 0 (vehicle), 150, 300, 600, 1200 mg/kg |
| ””Number of animals/group | : | Males, 5; females, 5 |
| ””Vehicle | : | 0.5 % Sodium carboxymethylcellulose |
| GLP | : | Yes |
””Test results:
The LD50 value was estimated to be 585.8 mg/kg for both sexes.
| Purity | : | > 99 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | Guideline for the 28-Day Repeated Dose Toxicity Test in Mammalian Species (Chemical Substances Control Law of Japan) |
| ””Route | : | Oral (gavage) |
| ””Dosage | : | 0 (vehicle), 30, 100, 300, 400 mg/kg |
| ””Number of animals/group | Males, 5; females, 5 | |
| ””Vehicle | : | 0.5 % Sodium carboxymethylcellulose |
| ””Administration period | : | Males and females, 28 days |
| ””Terminal killing | : | Males and females, days 29 or 43 |
| GLP | : | Yes |
””Test results:
On urinalysis, increase of urine volume was observed in males given 300 mg/kg, and males and females given 400 mg/kg. With 400 mg/kg, decrease of pH in males and females, and decrease of specific gravity in females were also observed. Water consumption was increased in males given 400 mg/kg.
On hematological examination, decrease of MCHC and prolongation of APTT were observed in females given 400 mg/kg. Decrease of MCHC was also observed in males given 300 and 400 mg/kg after the recovery period.
On biochemical examination, increase of ¦Ć-GTP was observed in males at 300 mg/kg and above, and females at 400 mg/kg. With 400 mg/kg, increase of ¦Ā-globulin in males and of ¦Į1-globulin in females was also observed.
Thickening of mucosa in the forestomach was observed in males and females given 400 mg/kg. Dilatation and watery contents of the cecum and atrophy of the spleen and thymus were observed in one female given 400 mg/kg.
Liver weights were increased in males given 100 mg/kg, and males and females given 300 and 400 mg/kg. Relative liver weight was increased in males given 300 mg/kg, and males and females given 400 mg/kg. Absolute and relative kidney weights were increased in males given 400 mg/kg. Absolute and relative thymus weights were decreased in females given 400 mg/kg.
Histopathological examination revealed squamous cell hyperplasia of the forestomach in females at 100 mg/kg and above. Squamous cell hyperplasia of the limiting ridge of the stomach was also observed in males and females at 100 mg/kg and above. Formation of hyaline droplets and eosinophilic bodies in the renal proximal tubular epithelium was observed in males given 400 mg/kg. Thymic atrophy was observed in females given 400 mg/kg.
Animals demonstrated reduction or disappearance of at the end of the 14 day recovery period.
The NOEL is considered to be 30 mg/kg/day for males and females.
| Purity | : | > 99 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 421 |
| ””Route | : | Oral (gavage) |
| ””Dosage | : | 0 (vehicle), 4, 15, 60 mg/kg/day |
| ””Number of animals/group | Males, 13; females, 13 | |
| ””Vehicle | : | 0.5 % Sodium carboxymethylcellulose |
| ””Administration period | : | Males, 47 days Females, from 14 days before mating to day 3 of lactation |
| ””Terminal killing | : | Males, day 48 of the administration Females, day 4 of lactation |
| GLP | : | Yes |
””Test results:
No deaths were observed of any animals. Temporary salivation after oral dosing was observed in males at 60 mg/kg. There were no changes related to the treatment with 4-(1-methylethenyl)phenol in terms of body weight and food consumption. Thickening of the mucosa in the forestomach accompanied by diffuse squamous hyperplasia was found in the 60 mg/kg group. A decrease in the thymus weight was observed in females of the 60 mg/kg group along with an increase in extramedullary hematopoiesis in the spleen.
There were no adverse effects on copulation, fertility and estrous cyclicity in any groups. In addition, no changes related to the treatment with 4-(1-methylethenyl)phenol were observed in gestation length, delivery and lactation. As for pups, there were no changes related to the treatment with 4-(1-methylethenyl)phenol in terms of morphology, body weight and viability.
The NOELs for systemic toxicity of 4-(1-methylethenyl)phenol was estimated to be 15 mg/kg/day in male and female rats, and that for reproductive and developmental toxicity 60 mg/kg/day.
| Purity | : | > 99 % |
| Test species/strain | : | Salmonella typhimurium TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 471 |
| ””Procedures | : | Pre-incubation method |
| ””Solvent | : | Dimethyl sulfoxide |
| ””Positive controls | : | -S9 mix; 2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide (TA100, TA98 and WP2 uvrA), Sodium azide (TA1535) and 9-Aminoacridine hydrochloride (TA1537) +S9 mix; 2-Aminoanthracene (all strains) |
| ””Dosage | : | -S9 mix; 0, 2.29, 6.86, 20.6, 61.7, 185, 556, 1667, 5000 ¦Ģg/plate (all strains for dose-finding study) +S9 mix; 0, 2.29, 6.86, 20.6, 61.7, 185, 556, 1667, 5000 ¦Ģg/plate (all strains for dose-finding study) -S9 mix; 0, 12.5, 25.0, 50.0, 100, 200, 400, 800 ¦Ģg/plate (TA100, TA1535, TA98 and TA1537 for main study) -S9 mix; 0, 50.0, 100, 200, 400, 800, 1600 ¦Ģg/plate (WP2 uvrA for main study) +S9 mix; 0, 12.5, 25.0, 50.0, 100, 200, 400, 800 ¦Ģg/plate (TA100, TA1535, TA98 and TA1537 for main study) +S9 mix; 0, 50.0, 100, 200, 400, 800, 1600 ¦Ģg/plate (WP2 uvrA for main study) -S9 mix; 0, 12.5, 25.0, 50.0, 100, 200, 400, 800 ¦Ģg/plate (TA100 for restudy) -S9 mix; 0, 50.0, 100, 200, 400, 800, 1600 ¦Ģg/plate (WP2 uvrA for restudy) |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
””Test results:
Genetic effects:
Salmonella typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
Escherichia coli WP2 uvrA
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | > 99 % |
| Type of cell used | : | Chinese hamster lung (CHL/IU) cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 473 |
| ””Solvent | : | Dimethyl sulfoxide |
| ””Positive controls | : | -S9 mix; Mitomycin C +S9 mix; Cyclophosphamide |
| ””Dosage | : | -S9 mix (short-term treatment); 0, 64.8, 108, 180 ¦Ģg/mL +S9 mix (short-term treatment); 0, 64.8, 108, 180 ¦Ģg/mL |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
””Test results:
| Lowest concentration producing cytogenetic effects in vitro | : | ||
| Without metabolic activation (short-term treatment) | : | 64.8 ¦Ģg/mL (clastogenicity) | |
| With metabolic activation (short-term treatment) | : | 108 ¦Ģg/mL (clastogenicity) | |
Genetic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| Without metabolic activation: | [*] | [ ] | [ ] | [ ] | [ ] | [*] |
| With metabolic activation: | [*] | [ ] | [ ] | [ ] | [ ] | [*] |
| 1) | The tests were performed by the Safety Research Institute for Chemical Compounds Co., Ltd., 363-24 Shin-ei, Kiyota-ku, Sapporo-shi, Hokkaido, 004-0839, Japan. Tel +81-11-885-5031 Fax +81-11-885-5313 |
| 2) | The test was performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257-8523, Japan. Tel +81-463-82-4751 Fax +81-463-82-9627 |
| 3) | The tests were performed by the Biosafety Research Center, Foods, Drugs and Pesticides (An-pyo Center), 582-2 Shioshinden, Fukude-cho, Iwata-gun, Shizuoka, 437-1213, Japan. Tel +81-538-58-1266 Fax +81-538-58-1393 |