
In the repeat dose toxicity test, body weight gain was suppressed in males and females given 200 and 600 mg/kg. Hematological examination revealed slight decreases in red blood cell counts, hematocrit values and hemoglobin concentrations in males given 600 mg/kg. Absolute liver weights were increased or tended to increase and relative liver weights were increased in males given 600 mg/kg and females given 200 mg/kg and more. Absolute weights of the right and left kidneys were increased or tended to be increased and relative kidney weights were increased in males given 67 mg/kg and more and in females given 600 mg/kg. Histopathological examination revealed slight hypertrophy of centrilobular hepatocytes in males and females given 600 mg/kg, increased incidences of hyaline droplets and eosinophilic bodies of the proximal tubular epithelium in males given 67 mg/kg and more, and slight regeneration of renal tubular epithelium in males given 200 and 600 mg/kg. On the basis of these findings, NOELs for repeat dose toxicity were considered to be less than 67 mg/kg/day for males, and 67 mg/kg/day for females, respectively.
With regard to reproductive ability, no effects were detected in males. Gestation length was prolonged, and number of corpora lutea and implantation sites decreased in females given 600 mg/kg. Numbers of pups born and those born alive were decreased in the 600 mg/kg group. On the basis of these findings, NOELs for reproductive/developmental toxicity were considered to be 600 mg/kg/day for males and 200 mg/kg/day for females and for offspring, respectively.
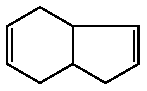
3a,4,7,7a-Tetrahydro-1H-indene was not mutagenic in Salmonella typhimurium TA100, TA1535, TA98, TA1537 and Escherichia coli WP2 uvrA, with or without an exogeneous metabolic activation system.
Genotoxicity of 3a,4,7,7a-tetrahydro-1H-indene was studied by chromosomal aberration test in cultured Chinese hamster lung (CHL/IU) cells. Structural chromosomal aberrations were induced at 0.10 mg/ml (high concentration) with short-term treatment and an exogenous metabolic activation system. Polyploidy was not induced in any treatment group.
| Purity | : | 99.0% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | OECD Combined Repeat Dose and Reproductive/Developmental Toxicity Screening Test |
| ””Route | : | Oral (gavage) |
| ””Dosage | : | 0 (vehicle), 67, 200 and 600 mg/kg/day |
| ””Number of animals | : | Males, 5; females, 5/group |
| ””Vehicle | : | Olive oil |
| ””Administration period | : | Males, 46 days Females, from 14 days before mating to day 3 of lactation |
| ””Terminal kill | : | Males, day 47 Females, day 4 of lactation |
| GLP | : | Yes |
””Test results:
| Purity | : | 99.0 wt% |
| Test species/strains | : | Salmonella typhimurium, TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) and OECD Guideline No. 471 and 472 |
| ””Procedures | : | Pre-incubation method |
| ””Solvent | : | DMSO |
| ””Positive controls | : | -S9 mix, 2-(2-Furyl)-3-(5-nitro-2-furyl) acrylamide (TA100, TA98, WP2), Sodium azide (TA1535) and 9-Aminoacridine (TA1537) +S9 mix, 2-Aminoanthracene (five strains) |
| ””Doses | : | -S9 mix; 0, 3.91, 7.81, 15.6, 31.3, 62.5, 125 ¦Ģg/plate (TA1535, TA1537) 0, 7.81 - 250 ¦Ģg/plate (TA100, TA98, WP2) +S9 mix; 0, 7.81 - 250 ¦Ģg/plate (TA1535, TA1537) 0, 15.6 - 500 ¦Ģg/plate (TA100, TA98, WP2) |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
””Test results:
Genetic effects:
Salmonella typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
Escherichia coli WP2 uvrA
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99.0% |
| Type of cell used | : | Chinese hamster lung (CHL/IU) cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) and OECD Guideline No. 473 |
| ””Solvent | : | Dimethylsulfoxide |
| ””Positive controls | : | -S9 mix, Mitomycin C +S9 mix, Cyclophosphamide |
| ””Doses | : | -S9 mix (continuous treatment):0, 0.013, 0.025, 0.050 mg/ml -S9 mix (short-term treatment):0, 0.013, 0.025, 0.050 mg/ml +S9 mix (short-term treatment):0, 0.025, 0.050, 0.10 mg/ml |
| ””S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
””Test results:
| With metabolic activation (short-term treatment) | :0.10 mg/ml (clastogenicity) |
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| With metabolic activation: | [*] | [ ] | [ ] | [ ] | [ ] | [*] |
| 1) | The tests were performed by the Safety Research Institute for Chemical Compounds Co., Ltd., 363-24, Shin-ei, Kiyota-ku, Sapporo, Hokkaido, 004, Japan. Tel +81-11-885-5031 Fax +81-11-885-5313 |
| 2) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257, Japan. Tel +81-463-82-4751 Fax +81-463-82-9627 |