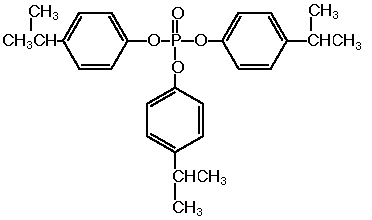

The single dose oral toxicity test revealed an LD50 of more than 2000 mg/kg for both sexes.
In the repeat dose study, the test substance caused salivation in males dosed with 40 mg/kg or more and in females dosed with 200 mg/kg or more, lower values of food consumption in females dosed with 200 mg/kg or more and in males of the 1000 mg/kg group, and higher water intake in females of the 1000 mg/kg group. The test substance also caused slight anemia and higher values for liver weights in both sexes of the groups dosed with 200 mg/kg or more, as well as increased urinary volume and serum total cholesterol, and lowered urinary specific gravity in females of the 1000 mg/kg group. All of these changes were reversible and disappeared within a 14-day recovery period. NOELs are considered to be 8 mg/kg/day for males and 40 mg/kg/day for females in terms of general toxicological effects.
Tris(p-cumenyl) phosphate was not mutagenic in Salmonella typhimurium TA100, TA98, TA1535, TA1537 and Escherichia coli WP2 uvrA. Neither structural nor numerical chromosomal aberrations were induced in CHL/IU cells up to the concentration going 50% cell growth inhibition or the concentration of 10 mM, in the absence or presence of an exogenous metabolic activation system.
| Purity | : | 99.7% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | OECD Test Guideline 401 |
| ””Dosage | : | 500, 1000, 2000 mg/kg |
| ””Number of animals | : | Male, 5 ; Female, 5/group |
| ””Vehicle | : | Corn oil |
| GLP | : | Yes |
| Purity | : | 99.7% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | Guidelines for 28-Day Repeat Dose Toxicity Testing of Chemicals (Japan) |
| ””Route | : | Oral (gavage) |
| ””Dosage | : | 8, 40, 200, 1000 mg/kg/day |
| ””Number of animals | : | Male, 10 or 15 (0, 1000 mg/kg) Female, 10 or 15 (0, 1000 mg/kg) |
| ””Vehicle | : | Corn oil |
| ””Administration period | : | Male, Female ; 28 days |
| ””Terminal kill | : | Male, Female ; Day 29 or 43 (0, 1000 mg/kg) |
| GLP | : | Yes |
NOELs are considered to be 8 mg/kg/day for males and 40 mg/kg for females in terms of general toxicological effects under the conditions of the present study.
| Purity | : | > 99% |
| Test species/strains | : | S.typhimurium TA100, TA1535, TA98, TA1537 E. coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ””Procedures | : | Plate incorporation method |
| ””Solvent | : | DMSO |
| ””Positive controls | : | -S9, AF-2 (TA100, WP2, TA98), sodium azide (TA1535) and 9-aminoacridine (TA1537) +S9, 2-aminoanthracene (all strains) |
| ””Dosage | : | 0, 312.5, 625, 1250, 2500, 5000¦Ģg/plate |
| ””S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
Genetic effects:
S. typhimurium TA100, TA1535, TA 98, TA1537
| + | ? | - | |
| with metabolic activation | [ ] | [ ] | [*] |
| without metabolic activation | [ ] | [ ] | [*] |
E. coli WP2 uvrA
| with metabolic activation | [ ] | [ ] | [*] |
| without metabolic activation | [ ] | [ ] | [*] |
| Purity | : | 99% |
| Type of cell used | : | Chinese hamster CHL/IU cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ””Solvent | : | DMSO |
| ””Positive controls | : | -S9, Mitomycin C +S9, Cyclophosphamide |
| ””Dosage | : | -S9 (continuous treatment): 0, 0.04, 0.07, 0.14 mg/ml -S9 (short-term treatment): 0, 1.1, 2.3, 4.5 mg/ml +S9 (short-term treatment): 0, 1.1, 2.3, 4.5 mg/ml |
| ””S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
Genotoxic effects:
| ”” | clastogenicity | polyploidy | ||||
| + | ? | - | + | ? | - | |
| without metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| with metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| 1) | The tests were performed by Nihon Bioresearch Inc. Hashima Laboratory, 6-104 Majima, Fukuju-cho, Hashima, 501-62, Japan. Tel +81-58-392-6222 Fax +81-58-392-1284 |
| 2) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa 257, Japan. Tel +81-463-82-4751 Fax +81-463-82-9627 |