
Death of one male occurred in the 300 mg/kg group which showed a decrease in spontaneous activity, adaption of a prone position and bradypnea. Two females in the same dose group showed the same clinical signs. The NOEL for repeat dose toxicity for both sexes is considered to be 100 mg/kg/day. No effects were observed on reproductive performance in either sex or on development of the next generation. NOELs for reproductive and developmental performances are considered to be 300 mg/kg/day.
3-Methyl-4-nitrophenol was not mutagenic in Salmonella typhimurium TA100, TA98, TA1535, TA1537 and Escherichia coli WP2 uvrA. This chemical substance induced structural chromosomal aberrations in CHL/IU cells, in the presence of an exogenous metabolic activation system. Numerical aberrations were not induced under the experimental conditions used.
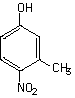
| Purity | : | 98.5% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | OECD Preliminary Reproductive/Developmental Toxicity Screening Test |
| ””Route | : | Oral (gavage) |
| ””Doses | : | 0 (vehicle), 30, 100, 300 mg/kg/day |
| ””Number of animals | : | Male, 12; female, 12/group |
| ””Vehicle | : | 10% gum arabic solution |
| ””Administration period | : | Male, 46 days
Female, from 14 days before mating to day 3 of lactation |
| ””Terminal kill | : | Male, day 47
Female, day 4 of lactation |
| GLP | : | Yes |
””Test results:
NOELs for reproductive and developmental performances are considered to be 300 mg/kg/day.
The NOEL for repeat dose toxicity for both sexes is considered to be 100 mg/kg/day.
| Purity | : | 99.9% |
| Test species/strains | : | S.typhimurium TA100, TA1535, TA98, TA1537, E. coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ””Procedures | : | Plate incorporation method |
| ””Solvent | : | DMSO |
| ””Positive controls | : | -S9, AF-2 (TA100, WP2, TA98), sodium azide (TA1535) and 9-aminoacridine (TA1537) +S9, 2-aminoanthracene (all strains) |
| ””Dosage | : | 0, 78.12, 156.2, 312.5, 625, 1250, 2500 ¦Ģg/plate |
| ””S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
| + | ? | - | |
| with metabolic activation | [ ] | [ ] | [*] |
| without metabolic activation | [ ] | [ ] | [*] |
| with metabolic activation | [ ] | [ ] | [*] |
| without metabolic activation | [ ] | [ ] | [*] |
| Purity | : | 99.9% |
| Type of cell used | : | Chinese hamster CHL/IU cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ””Solvent | : | DMSO |
| ””Positive controls | : | -S9, Mitomycin C +S9, Cyclophosphamide |
| ””Doses | : | -S9 (continuous treatment): 0, 0.006, 0.012, 0.023 mg/ml
-S9 (short-term treatment): 0, 0.04, 0.08, 0.15 mg/ml +S9 (short-term treatment): 0, 0.04, 0.08, 0.15 mg/ml |
| ””S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| without metabolic activation: | [ ] | [*] | [ ] | [ ] | [ ] | [*] |
| with metabolic activation: | [*] | [ ] | [ ] | [ ] | [ ] | [*] |
| 1) | The test was performed by the Safety Research Institute for Chemical Compound Co., Ltd., 363-24, Shin-ei, Toyohira-ku, Sapporo, Hokkaido 004, Japan. Tel +81-11-885-5031 Fax +81-11-885-5313 |
| 2) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa 257, Japan. Tel +81-463-82-4751 Fax +81-463-82-9627 |