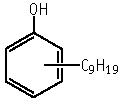

Nonylphenol was not mutagenic to Salmonella typhimurium, TA100, TA1535, TA98, TA1537 and Escherichia coli WP2 uvrA, with or without an exogeneous metabolic activation system.
Nonylphenol induced neither structural chromosomal aberrations nor polyploidy in CHL/IU cells, in the absence or presence of an exogenous metabolic activation system.
| Purity | : | 99.0 % |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | Guidelines for 28-Day Repeat Dose Toxicity Testing of Chemicals (Japan) |
| Route | : | Oral (gavage) |
| Doses | : | 0 (vehicle), 4, 15, 60, 250 mg/kg/day |
| Number of animals/group | : | Males, 6; females, 6 |
| Vehicle | : | Olive oil |
| Administration period | : | Males and females, 28 days |
| Terminal kill | : | Days 29 or 43 |
| GLP | : | Yes |
Test results:
| Purity | : | > 99.0% |
| Test species/strain | : | S. typhimurium TA100, TA1535, TA98, TA1537, E. coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| Procedures | : | Pre-incubation method |
| Solvent | : | DMSO |
| Positive controls | : | -S9 mix AF-2 (TA100, TA98 and WP2 uvrA) Sodium azide (TA1535) 9-Aminoacridine (TA1537) +S9 mix 2-Aminoanthracene (all strains) |
| Doses | : | -S9 mix (TA100, TA98, TA1535 and TA1537) 0.195, 0.391, 0.781, 1.56, 3.13, 6.25, 12.5 μg/plate -S9 mix (WP2 uvrA) 1.56, 3.13, 6.25, 12.5, 25.0, 50.0 μg/plate +S9 mix (TA100, TA1535 and TA1537) 1.56, 3.13, 6.25, 12.5, 25.0, 50.0 μg/plate +S9 mix (TA98 and WP2 uvrA) 6.25, 12.5, 25.0, 50.0, 100, 200 μg/plate |
| S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| Plates/test | : | 3 |
| Number of replicates | : | 2 |
| GLP | : | Yes |
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | > 99.0% |
| Type of cell used | : | Chinese hamster lung (CHL) cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| Solvent | : | DMSO |
| Positive controls | : | long-term treatment; Mitomycin C (24 h and 48 h) short-term treatment; Cyclophosphamide (+S9 mix and -S9 mix) |
| Doses | : | long-term treatment; 6.25, 12.5, 25.0, 50.0 μg/ml (24 h) long-term treatment; 3.13, 6.25, 12.5, 25.0 μg/ml (48 h) short-term treatment; 7.50, 15.0, 30.0, 60.0 μ g/ml (+S9 mix and -S9 mix) |
| S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| Plates/test | : | 2 |
| GLP | : | Yes |
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| without metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| with metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| 1) | The tests were performed by Bozo Research Center Inc, 1284, Kamado, Gotemba-shi, Shizuoka, 412, Japan. Tel +81-550-82-2000 Fax +81-550-82-2379 |
| 2) | The tests were performed by the Biosafety Research Center, Foods, Drugs and Pesticides (An-pyo Center), Japan, 582-2 Shioshinden Arahama, Fukude-cho, Iwata-gun, Shizuoka, 437-12, Japan. Tel +81-538-58-1266 Fax +81-538-58-1393 |