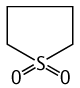

Tetrahydrothiophene-1,1-dioxide was studied for oral toxicity in rats in an OECD preliminary reproduction toxicity screening test at doses of 0, 60, 200, and 700mg/kg/day. One animal of each sex died in the 700 mg/kg group (12 animals of each sex treated).
In the males, regarding general signs, soiled fur, diarrhea, and soft stools were noted in the 700 mg/kg group. Suppression of body weight gain was also noted in this group and food consumption was decreased. In the females, regarding general signs, soiled fur was noted in the 700 mg/kg group, together with suppression of body weight gain during the pre-mating period. Food consumption decreased in the 700 mg/kg group during the pre-mating and lactation periods. The relative ovary weights were increased in the 700 mg/kg group. The NOELs for repeat dose toxicity are considered to be 200 mg/kg/day for both sexes.
With regard to reproductive/developmental toxicity, the number of estrous cases decreased in the 700 mg/kg group. Four dams in the 700 mg/kg group lost all their pups during the lactation period. The NOELs for reproductive performance are considered to be 700 mg/kg/day for males, and 200 mg/kg/day for females.
With regard to pups, birth index, live birth index, number of pups on days 0 and 4 of lactation, viability index, and body weights of both sexes on days 0 and 4 of lactation were decreased, and the number of stillbirths increased in the 700 mg/kg group. Birth index and the number of pups on days 0 and 4 of lactation decreased in the 200 mg/kg group. The NOEL for pup development is considered to be 60 mg/kg/day.
| Purity | : | 97.3 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Preliminary Reproduction Toxicity Screening Test |
| Route | : | Oral (gavage) |
| Doses | : | 0 (vehicle), 60, 200, 700 mg/kg/day |
| Number of animals/group | : | Males, 12; females, 12 |
| Vehicle | : | Water for injection |
| Administration period | : | Males, 49 days Females, from 14 days before mating to day 3 of lactation |
| Terminal kill | : | Males, day 50 Females, day 4 of lactation |
| GLP | : | Yes |
Test results:
For the males, soiled fur, diarrhea, soft stool, suppression in body weight gain, and a decrease in food consumption were noted, and 1 of the 12 died in the 700 mg/kg group.
For the females, soiled fur, suppression in body weight gain during the pre-mating period, decreases in food consumption during the pre-mating and lactation periods, and an increase in the relative ovary weight were noted, and 1 of the 12 died in the 700 mg/kg group.
The NOELs for repeat dose toxicity are considered to be 200 mg/kg/day for both sexes.
<Reproductive and developmental toxicity>
With regard to reproductive/developmental toxicity, the number of estrous cases was decreased in the 700 mg/kg group. Four dams lost all their pups during the lactation period in the 700 mg/kg group. The NOELs for reproductive performance are considered to be 700 mg/kg/day for males, and 200 mg/kg/day for females.
With regard to pups, birth index, live birth index, number of pups on days 0 and 4 of lactation, viability index, and body weights of both sexes on days 0 and 4 of lactation were decreased, and the number of stillbirths increased in the 700 mg/kg group. Birth index and the number of pups on days 0 and 4 of lactation decreased in the 200 mg/kg group. The NOEL for pup development is considered to be 60 mg/kg/day.
| 1) | The test was performed by Nihon Bioresearch Inc., Hashima Laboratory, 6-104 Majima, Fukuju-cho, Hashima, Gifu, 501-6251, Japan. Tel +81-58-392-6222 Fax +81-58-392-1284 |