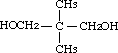

2,2-Dimethyl-1,3-propanediol was studied for oral toxicity in rats in an OECD combined repeat dose and reproductive/developmental toxicity screening test at doses of 0, 100, 300 and 1000 mg/kg/day, and for mutagenicity with assays for reverse mutation in bacteria and chromosomal aberrations in cultured Chinese hamster (CHL) cells.
For repeat dose toxicity, blood chemical examinations revealed increases in levels of total protein, total bilirubin and albumin among males given 300 and 1000 mg/kg, and a decrease in glucose level in the 1000 mg/kg dose males. Absolute and relative weights of the liver and kidneys were elevated in the 300- and 1000 mg/kg-dose males. Moreover, absolute and relative kidney weights were elevated in the 1000 mg/kg-dose males and demonstrated a tendency for increase in the 300 mg/kg dose males. Necropsy revealed hypertrophy of the liver in males given 1000 mg/kg. Histopathological examination showed increases in protein casts, hyaline degeneration and basophilic changes of the renal tubular epithelial cells in males given 1000 mg/kg. NOELs for repeat dose toxicity were 100 mg/kg/day in males and 1000 mg/kg /day in females. The compound showed no reproductive/developmental toxicity. NOELs for reproductive performance and offspring development were both 1000 mg/kg/day.
The mutagenicity tests were all negative. 2,2-Dimethyl-1,3-propanediol was not mutagenic for bacteria either with or without exogenous metabolic activation up to 5000 ŠÌg/plate. Neither chromosomal aberrations nor polyploidy were induced in CHL cells up to 1.0 mg/ml with or without exogenous metabolic activation.
| Purity | : | 99.15% |
| Test species/strain | : | Rat/Slc:SD |
| Test method | : | OECD Combined Repeat Dose and Reproductive/Developmental Toxicity Screening Test |
| ĄĄRoute | : | Oral (gavage) |
| ĄĄDoses | : | 0 (vehicle), 100, 300, 1000 mg/kg/day |
| ĄĄNumber of animals | : | Male, 12; Female, 12/group |
| ĄĄVehicle | : | Distilled water |
| ĄĄAdministration period | : | Male, 45 days Female, from 14 days before mating to day 3 of lactation |
| ĄĄTerminal kill | : | Male, day 46 Female, day 4 of lactation |
| ĄĄGLP | : | Yes |
ĄĄTest results:
There were no observed effects of the test substance on hematological findings for the treated male rats. Blood chemical examinations revealed elevations in levels of total protein, total bilirubin and albumin among male rats receiving 300 and 1000 mg/kg and a decrease in glucose in male rats receiving 1000 mg/kg.
Absolute and relative weights of the liver and kidneys of male rats receiving 300 and 1000 mg/kg were elevated. Absolute and relative kidney weights were elevated in male rats receiving 1000 mg/kg. A non-significant tendency for increase was also evident in males receiving 300 mg/kg. Necropsy revealed hypertrophy of the liver in 2/12 male rats receiving 1000 mg/kg. However, histological examination revealed no definite associated lesion. Histopathological examination revealed increases in the amounts of protein casts, hyaline degeneration and basophilic alteration of the renal tubular epithelium in male rats receiving 1000 mg/kg.
| NOELs: | Male 100 mg/kg/day Female 1000 mg/kg/day |
<Reproductive and Developmental toxicity>
There were no effects of the test substance on copulation, fertility or estrus cycle of rats. Delivery was normal for dams except for one animal of the control group. No effects of the test substance were observed on dams during the lactation period. External examination of pups revealed no increase of abnormalities due to thetest substance. Body weight gain of the pups was normal up to day 4 of the lactation period. Stillborn, dead pups and pups killed at day 4 of the lactation period showed no abnormal gross findings attributable to treatment with the test substance.
| Purity | : | 99.15% |
| Test species/strains | : | S. typhimurium TA100, TA1535, TA98, TA1537 E. coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ĄĄProcedure | : | Plate method |
| ĄĄSolvent | : | Distilled water |
| ĄĄPositive controls | : | -S9, AF-2 (TA100,WP2 uvrA,TA98), sodium azide (TA1535) and 9-aminoacridine (TA1537) +S9, 2-Aminoanthracene (all strains) |
| ĄĄDoses | : | 0, 312.5, 625, 1250, 2500, 5000 ŠÌg/plate |
| ĄĄS-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ĄĄPlates/test | : | 3 |
| ĄĄNumber of replicates | : | 2 |
| GLP | : | Yes |
Genotoxic effects:
S. typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| with metabolic activation: | [ ] | [ ] | [*] |
| without metabolic activation: | [ ] | [ ] | [*] |
E. coli WP2 uvrA
| + | ? | - | |
| with metabolic activation: | [ ] | [ ] | [*] |
| without metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99.15% |
| Type of cell used | : | Chinese hamster CHL cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ĄĄSolvent | : | Distilled water |
| ĄĄPositive controls | : | Mitomycin C Cyclophosphamide |
| ĄĄDoses | : | 0, 0.25, 0.5, 1 mg/ml |
| ĄĄS-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ĄĄPlates/test | : | 2 |
| GLP | : | Yes |
| + | ? | - | |
| with metabolic activation: | [ ] | [ ] | [*] |
| without metabolic activation: | [ ] | [ ] | [*] |
| 1) | The test was performed by the Biosafety Research Center, Foods, Drugs and Pesticides (An-pyo Center), 582-2 Shioshinden Arahama, Fukude-cho, Iwata-gun, Shizuoka, 437-12, Japan. Tel 81-538-58-1266 Fax 81-538-58-1393 |
| 2) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257, Japan. Tel 81-463-82-4751 Fax 81-463-82-9627 |