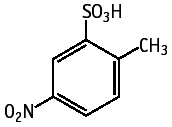

2-Methyl-5-nitrobenzenesulfonic acid was studied for oral toxicity in rats an OECD combined repeat dose and reproductive/developmental toxicity screening test at doses of 0, 175, 350 and 700 mg/kg/day.
With regard to repeat dose toxicity, hyperplasia of mucosa of the limiting ridge of the stomach and atrophy of mucosa in the cardiac region were observed in males and females of the 350 and 700 mg/kg groups. Erosion and superficial hemorrhage in the glandular stomach were also observed in males and females of the 700 mg/kg group, along with wheezing, soft feces with partial black changes and abdominal distension. Further, retarded body weight gain, low food consumption, low values for mean corpuscular volume and mean corpuscular hemoglobin concentration and low values for total protein and ¦Į1-globulin fraction ratio were observed in males of the 700 mg/kg group. In the females, one animal died and one animal was sacrificed on becoming moribund, and retarded body weight gain was observed in the 700 mg/kg group. The NOEL for repeat dose toxicity is considered to be 175 mg/kg/day for males and females.
With regard to reproductive/developmental toxicity, there were no effects of the test substance in the 700 mg/kg group. As for pups, tendencies toward low values for body weights of males and females and for viability on day 4 were observed in the 700 mg/kg group.
The NOELs for the reproductive/developmental toxicity are considered to be 700 mg/kg/day for reproduction for males and females, and 350 mg/kg/day for pups.
2-Methyl-5-nitrobenzenesulfonic acid was mutagenic in Salmonella typhimurium TA100, TA98 and TA1537 without an exogenous metabolic activation system, and in TA100 with an exogenous metabolic activation system.
2-Methyl-5-nitrobenzenesulfonic acid did not induce structural chromosomal aberrations or polyploidy in CHL cells, with or without an exogenous metabolic activation system.
| Purity | : | 79.6 % |
| Test Species/Strain | : | Rat/Crj:CD(SD) |
| Test method | : | OECD Combined Repeat Dose and Reproductive/ Developmental Toxicity Screening Test |
| ””Route | : | Oral(gavage) |
| ””Dose | : | 0(Vehicle), 175, 350 and 700 mg/kg/day |
| ””Number of animals/group | : | Males, 12; females, 12 |
| ””Vehicle | : | Water for injection |
| ””Administration period | : | Males, 49 or 50 days Females, from 14 days before mating to the day before autopsy(day 3 of lactation) |
| ””Terminal sacrifice | : | Males, days 50 or 51 of administration Females, day 4 of lactation |
| GLP | : | Yes |
””Test results:
In males of the 700 mg/kg group, wheezing, soft feces with partial black changes, abdominal distension, soiling of the peri-anal area and unkempt fur, retarded body weight gain, and low food consumption were observed. In addition, low values for mean corpuscular volume and mean corpuscular hemoglobin and low values for total protein and ¦Į1-globulin fraction ratio were observed. Further, gross pathologically, thickening of the limiting ridge, dark red spots and erosion in the glandular stomach were seen, and histopathologically, hyperplasia of mucosa at the limiting ridge, atrophy of mucosa in the cardiac region, and erosion and superficial hemorrhage in the glandular stomach were observed. There were no effects of the test substance on myelogram findings, organ weights or staging of the seminiferous tublues. In females of the 700 mg/kg group, in addition to the changes observed in the males, paleness of pinna, emaciation, piloerection, decreased spontaneous movement, hypothermia and oligopnea were seen, and one female died and one female was sacrificed on becoming moribund. Further, a tendency toward retarded body weight gain was noted. Thickening of the limiting ridge, disseminated red spots and recessed areas in the glandular stomach, and hyperplasia of mucosa in the limiting ridge, atrophy of mucosa in the cardiac region, erosion and superficial hemorrhage in the glandular stomach were seen. There were no effects of the test substance on food consumption. In the 350 mg/kg group, gross thickening at the limiting ridge was seen in males, and histopathologically, hyperplasia of mucosa at the limiting ridge and atrophy of mucosa in the cardiac region were seen in males and females. There were no effects of the test substance on clinical signs, body weights, food consumption and hematology, myelogram or blood chemistry findings. In males and females of the 175 mg/kg group, there were no effects of the test substance on any parameter assessed.
The NOEL for repeat dose toxicity is considered to be 175 mg/kg/day for males and females.
<Reproductive/developmental toxicity>
With regard to male and female parents, there were no effects of the test substance on the estrous cycle, copulation index, fertility index, length of gestation, delivery and lactation conditions, the number of corpora lutea and implantation sites, implantation index, birth index or delivery index.
With regard to pups, tendencies toward low body weights in males and females and toward low viability on day 4 were observed in the 700 mg/kg group. However there were no effects of the test substance on the number of live born pups, number of stillborn pups, the live birth index, sex ratio, external anomalies or gross pathology.
The NOELs for the reproductive/developmental toxicity are considered to be 700 mg/kg/day for parental males and females, and 350 mg/kg/day for pups.
| Purity | : | 79.60 % |
| Test species/strains | : | S. typhimurium TA100, TA1535, TA98, TA1537, E. coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan)and OECD Guidelines No. 471 and 472 |
| ””Procedures | : | Pre-incubation method |
| ””Solvent | : | Distilled water |
| ””Positive controls | : | -S9 mix; 2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide(TA100, TA98 and WP2 uvrA), Sodium azide(TA1535), 9-Aminoacridine hydrochloride(TA1537) +S9 mix; 2-Aminoanthracene(all strains) |
| ””Doses | : | -S9 mix; 156, 313, 625, 1250, 2500 and 5000 ¦Ģg/plate +S9 mix; 156, 313, 625, 1250, 2500 and 5000 ¦Ģg/plate -S9 mix(TA1535; confirmative examination); 1000, 2000, 3000, 4000 and 5000 ¦Ģg/plate +S9 mix(TA1535; confirmative examination); 1000, 2000, 3000, 4000 and 5000 ¦Ģg/plate |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
””Test results:
Genetic effects:
S. typhimurium TA100
| + | ? | - | |
| Without metabolic activation: | [*] | [ ] | [ ] |
| With metabolic activation: | [*] | [ ] | [ ] |
S. typhimurium TA98 and TA1537
| + | ? | - | |
| Without metabolic activation: | [*] | [ ] | [ ] |
| With metabolic activation: | [ ] | [ ] | [*] |
S. typhimurium TA1535
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
E. coli WP2 uvrA
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 79.60 % |
| Type of cell used | : | Chinese hamster lung(CHL)cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan)and OECD Guideline No. 473 |
| ””Solvent | : | Saline |
| ””Positive controls | : | -S9 mix, Mitomycin C +S9 mix, Cyclophosphamide |
| ””Doses | : | -S9 mix(continuous exposure): 0, 550, 1100, 2200 ¦Ģg/mL -S9 mix(short-term exposure): 0, 550, 1100, 2200 ¦Ģg/mL +S9 mix(short-term exposure): 0, 550, 1100, 2200 ¦Ģg/mL |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
””Test results:
Genetic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| 1) | The tests were performed by Bozo Research Center Inc, 1284, Kamado, Gotemba-shi, Shizuoka, 412-0039, Japan. Tel +81-550-82-2000 Fax +81-550-82-2379 |
| 2) | The tests were performed by the Biosafety Research Center, Foods, Drugs and Pesticides(An-pyo Center), Japan, 582-2 Shioshinden Arahama, Fukude-cho, Iwata-gun, Shizuoka, 437-1213, Japan. Tel +81-538-58-1266 Fax +81-538-58-1393 |