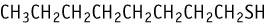
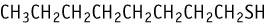
1-Octanethiol was studied for oral toxicity in rats in an OECD combined repeat dose and reproductive/developmental toxicity screening test at doses of 0, 10, 50, and 250 mg/kg/day.
As main effects of repeated dose toxicity, in the 250 mg/kg group, decreases in locomotor activity and salivation, and suppression of body weight gain were observed in both sexes. Hematological examination revealed decreases in the red blood cell count and MCHC, and increases in MCV and reticulocytes in the 250 mg/kg group in both sexes. Histopathological examination revealed increases in hematopoietic cells and hemosiderin deposition in the spleen and bone marrow in the 250 mg/kg group, and irritative changes of the stomach in the 50 mg/kg or higher groups. The NOEL for repeated dose toxicity is considered to be 10 mg/kg/day for both sexes.
Regarding reproductive and developmental toxicity, the test substance was associated with extension of the estrous cycle and a low delivery index in females of the 250 mg/kg group. The test substance had no effects on other parameters. The NOELs for reproductive and developmental toxicity are considered to be 250 mg/kg/day for males, 50 mg/kg/day for females, and 250 mg/kg/day for offspring.
1-Octanethiol was not mutagenic in Salmonella typhimurium TA100, TA1535, TA98, TA1537 or Escherichia coli WP2 uvrA/pKM101, with or without an exogenous metabolic activation system.
1-Octanethiol did not induce structural chromosomal aberrations or polyploidy in CHL/IU cells, with or without exogenous metabolic activation.
| Purity | : | 99.3 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 401 |
| ””Route | : | Oral (Gavage) |
| ””Dosage | : | 0 (Vehicle), 250, 500, 1000, 2000 mg/kg |
| ””Number of animals/group | : | Males, 5; females, 5 |
| ””Vehicle | : | Olive oil |
| GLP | : | Yes |
””Test results:
Clinical signs were noted as follows: decrease in locomotor activity, ataxic gait, abnormal gait, adoption of a lateral, prone or crouching position, lacrimation, reddish tears, smudging of the perinasal area, a soiled perineal region, anemic symptoms, hypothermia, tremors, irritability, and salivation. Furthermore, suppression of body weight gain was observed in the 500, 1000, and 2000 mg/kg groups. Necropsy revealed erosion/ulceration and white patches in the forestomach, focal hemorrhage and thickening of the wall in the glandular stomach, tar-like contents in the stomach and small intestine, duodenal ulceration, discoloration, interlobar adhesion and black patches in the liver, brown patches in the kidney, and retention of hematuria in the urinary bladder among the animals which died, as well as white and raised patches in the forestomach, enlargement and dark reddish change in the spleen, and adhesion of the liver and duodenum among the surviving animals. Histological examination revealed ulceration and inflammatory cell infiltration in the forestomach, erosion in the glandular stomach, hydropic degeneration of hepatocytes in the liver, and hemorrhage of tubules and necrosis of proximal tubular epithelium in the kidneys among the animals which died, as well as ulceration, erosion, inflammatory cell infiltration, and squamous cell hyperplasia in the forestomach, granulation tissue in the stomach, and erythrocytic extramedullary hematopoiesis in the spleen among the surviving animals.
The LD50 values were found to be about 2000 mg/kg for males and 1293 mg/kg for females.
| Purity | : | 99.3 % |
| Test species/strain | : | Rats/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 422 |
| ””Route | : | Oral (Gavage) |
| ””Dosage | : | 0 (Vehicle), 10, 50, 250 mg/kg |
| ””Number of animals/group | : | Males, 12; females, 12 |
| ””Vehicle | : | Olive oil |
| ””Administration period | : | Males, 35 days Females, from 14 days before mating to day 4 of lactation |
| ””Terminal killing | : | Male, day 36 Female, day 5 of lactation |
| GLP | : | Yes |
””Test results:
1-Octanethiol was studied for oral toxicity in rats in an OECD combined repeat dose and reproductive/developmental toxicity screening test at doses of 0, 10, 50, and 250 mg/kg/day.
No deaths occurred in any of the treated groups. In the 250 mg/kg group, decreases in locomotor activity and salivation were observed, and there was suppression of body weight gain in both sexes. Hematological examination in the 250 mg/kg group revealed decreases in the red blood cell count and MCHC, and increases in MCV and reticulocytes in both sexes. In addition, there was a decrease in hemoglobin concentration in males and an increase in MCH in females. Blood chemistry examination in the 250 mg/kg group revealed increases in albumin, the A/G ratio and inorganic phosphorus in both sexes, and revealed a decrease in Cl in males, and increases in GPT and total bilirubin, and a decrease in triglycerides in females. As for organ weight, there were increases in spleen, liver and kidney weights and a decrease in thymus weights in the 250 mg/kg group of both sexes. As necropsy findings, there were dark reddening and/or enlargement in the spleen in the 250 mg/kg group of both sexes, and thickening of the forestomach wall in the 50 mg/kg or higher groups in both sexes. Histopathological examination revealed increases in hematopoietic cells and hemosiderin deposition in the spleen and bone marrow in the 250 mg/kg group, and the irritative changes of stomach in the 50 mg/kg and higher groups.
The NOEL for repeated dose toxicity is considered to be 10 mg/kg/day for both sexes.
<Reproductive and developmental toxicity>
In the 250 mg/kg group, extension of the estrous cycle and a low delivery index were observed in female parental animals. The compound had no effects on other reproductive parameters such as the mating index, the fertility index, numbers of corpora lutea or implantations, the implantation index, the gestation index, gestation length, parturition or maternal behavior. On examination of neonates, there was no significant variation in numbers of offspring or live offspring, the sex ratio, the live birth index, viability index or body weight. No abnormal findings ascribable to the compound were found for external or clinical signs, of necropsy of the offspring.
The NOELs for reproductive and developmental toxicity are considered to be 250 mg/kg/day for males, 50 mg/kg/day for females, and 250 mg/kg/day for offspring.
| Purity | : | 99.3 % |
| Test species/strain | : | Salmonella typhimurium TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA/pKM101 |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 471 |
| ””Procedures | : | Pre-incubation method |
| ””Solvent | : | Dimethyl sulfoxide |
| ””Positive controls | : | -S9 mix; 2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide (TA100, TA98), Sodium azide (TA1535), 9-Aminoacridine hydrochloride (TA1537) and N-Ethyl-N'-nitro-N-nitro-soguanidine (WP2 uvrA/pKM101) +S9 mix; 2-Aminoanthracene (all strains) |
| ””Dosage | : | -S9 mix; 0, 0.610, 1.22, 2.44, 4.88, 9.77, 19.5 ¦Ģg/plate (TA100, TA98, TA1537) -S9 mix; 0, 0.153, 0.305, 0.610, 1.22, 2.44, 4.88, 9.77 ¦Ģg/plate (TA1535) -S9 mix; 0, 0.610, 1.22, 2.44, 4.88, 9.77, 19.5, 39.1 ¦Ģg/plate (WP2 uvrA/pKM101) +S9 mix; 0, 2.44, 4.88, 9.77, 19.5, 39.1, 78.1, 156 ¦Ģg/plate (TA100, TA1535, TA98, TA1537) +S9 mix; 0, 9.77, 19.5, 39.1, 78.1, 156, 313, 625 ¦Ģg/plate (WP2 uvrA/pKM101) |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
””Test results:
Genetic effects:
Salmonella typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
Escherichia coli WP2 uvrA/pKM101
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99.3 % |
| Type of cell used | : | Chinese hamster CHL/IU cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 473 |
| ””Solvent | : | Acetone |
| ””Positive controls | : | -S9 mix; Mitomycin C +S9 mix; Benzo[a]pyrene |
| ””Dosage | : | -S9 mix(6 hr short-term treatment); 0, 78.1, 156, 313, 625, 1250 ¦Ģg/mL +S9 mix(6 hr short-term treatment); 0, 625, 1250, 2500, 5000 ¦Ģg/mL -S9 mix(24 hr continuous treatment); 0, 70, 80, 90, 100, 110, 120 ¦Ģg/mL |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
””Test results:
Genotoxic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| 1) | The tests were performed by the Mitsubishi Chemical Safety Institute Ltd., 14 Sunayama, Hasaki-machi, Kashima-gun, Ibaraki, 314-0255, Japan. Tel +81-479-46-2871 Fax +81-479-46-2874 |