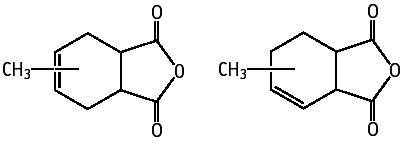

The compound was also studied for oral toxicity in Crj:CD (SD) rats in an OECD combined repeat dose and reproductive/developmental toxicity screening test at doses of 0, 30, 100 and 300 mg/kg/day.
With regard to repeated dose toxicity, salivation, decreased total cholesterol and BUN, increased triglyceride and increased adrenal weights were observed in males of the 300 mg/kg group. Histopathological examination revealed squamous hyperplasia of the forestomach in males of the 100 mg/kg group and both sexes of the 300 mg/kg group, submucosal granulomatous inflammation of the forestomach in both sexes of the 300 mg/kg group, epithelial vacuolar change, edema and cellular infiltration of the forestomach in males of the 300 mg/kg group, and erosion of the forestomach in females of the 300 mg/kg group. The NOELs for repeat dose toxicity are considered to be 30 mg/kg/day for males and 100 mg/kg/day for females. With regard to reproductive/developmental toxicity, there were no effects related to the test article on reproductive performance of parents and development of the next generation. The NOEL is considered to be more than 300 mg/kg/day for reproductive performance of parents and for development of offspring.
Tetrahydromethyl-1,3-isobenzofuranedione was not mutagenic in Salmonella typhimurium TA100, TA1535, TA98, TA1537 and Escherichia coli WP2 uvrA, with or without an exogeneous metabolic activation system.
Genotoxicity of tetrahydromethyl-1,3-isobenzofuranedione was studied by chromosomal aberration test in cultured Chinese hamster lung (CHL/IU) cells. Structural chromosomal aberrations were not induced. Polyploidy was observed with 0.30 mg/ml 48 h continuous treatment and with 0.11-0.43 mg/ml (all concentrations) short-term treatment with an exogenous metabolic activation system.
| Purity | : | 99.97% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | OECD Test Guideline 401 |
| Route | : | Oral (gavage) |
| Doses | : | 0 (Vehicle), 500, 1000, 2000 mg/kg/day |
| Number of animals/group | : | Males, 5 ; females, 5 |
| Vehicle | : | Corn oil |
| GLP | : | Yes |
Test results:
LD50 : Male, >2000 mg/kg ; female, >2000 mg/kg
| Purity | : | 99.97% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | OECD Combined Repeat Dose and Reproductive/Developmental Toxicity Screening Test |
| Route | : | Oral (gavage) |
| Dosage | : | 0 (Vehicle), 30, 100, 300 mg/kg/day |
| Number of animals/group | : | Males, 12 ; females, 12 |
| Vehicle | : | Corn oil |
| Administration period | : | Males, 49 days Females, from 14 days before mating to day 3 of lactation |
| Terminal kill | : | Males, day 50 Females, day 4 of lactation |
| GLP | : | Yes |
Test results:
| Purity | : | 99.97 wt% |
| Test species/strains | : | Salmonella typhimurium, TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) and OECD Guideline No. 471 and 472 |
| Procedures | : | Pre-incubation method |
| Solvent | : | DMSO |
| Positive controls | : | -S9 mix, 2-(2-Furyl)-3-(5-nitro-2-furyl) acrylamide (TA100, TA98, WP2), Sodium azide (TA1535) and 9-Aminoacridine (TA1537) +S9 mix, 2-Aminoanthracene (five strains) |
| Doses | : | -S9 mix; 0, 62.5, 125, 250, 500, 1000, 2000 μg/plate (TA100, TA1535, TA98, TA1537) 0, 156 - 5000 μg/plate (WP2) +S9 mix; 0, 313 - 5000 μg/plate (five strains) |
| S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| Plates/test | : | 3 |
| Number of replicates | : | 2 |
| GLP | : | Yes |
Test results:
Genetic effects:
Salmonella typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
Escherichia coli WP2 uvrA
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99.97% |
| Type of cell used | : | Chinese hamster lung (CHL/IU) cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) and OECD Guideline No. 473 |
| Solvent | : | Dimethylsulfoxide |
| Positive controls | : | -S9 mix, Mitomycin C +S9 mix, Cyclophosphamide |
| Doses | : | -S9 mix (continuous treatment):0, 0.075, 0.15, 0.30 mg/ml -S9 mix (short-term treatment):0, 0.05, 0.10, 0.20 mg/ml +S9 mix (short-term treatment):0, 0.11, 0.21, 0.43 mg/ml |
| S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| Plates/test | : | 2 |
| GLP | : | Yes |
Test results:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] | [ ] | [*] | [ ] |
| With metabolic activation: | [ ] | [ ] | [*] | [ ] | [*] | [ ] |
| 1) | The tests were performed by Safety Assessment Laboratory, Panapharm Laboratories Co., Ltd. 1285 Kurisaki-machi, Uto-shi, Kumamoto, 869-04, Japan Tel +81-964-23-5111 Fax +81-964-23-2282 |
| 2) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257, Japan. Tel +81-463-82-4751 Fax +81-463-82-9627 |