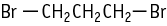
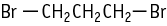
1,3-Dibromopropane was studied for oral toxicity in rats in a 28-day repeated dose toxicity test at doses of 0, 10, 50 and 250 mg/kg.
Salivation, anemic changes (occurring in the recovery period in males), increases in total protein and lipid, increased liver weight, hypertrophy of the centrilobular hepatocytes and decreased vacuolation in the perilobular hepatocytes were observed in both sexes given 250 mg/kg. Of these, hypertrophy of the centrilobular hepatocytes in both sexes and increased liver weight, decreased vacuolation in the perilobular hepatocytes and an increase in total protein were also observed in males given 50 mg/kg. No treatment-related changes were noted in the 10 mg/kg group. Therefore, the NOEL is considered to be 10 mg/kg/day for both sexes.
Reverse mutation assays using microorganisms (Salmonella typhimurium TA100, TA1535, TA98, TA1537 and Escherichia coli WP2 uvrA) were conducted to assess the potential of 1,3-dibromopropane to induce gene mutations. Mutagenic activity was found in S. typhimurium TA100 and TA1535 with metabolic activation under the present experimental conditions.
In vitro tests using cultured Chinese hamster cells (CHL/IU) were conducted to assess the potential of 1,3-dibromopropane to induce chromosomal aberrations. Structural chromosome aberrations were induced under the present experimental conditions, suggesting that the test substance, 1,3-dibromopropane, is clastogenic to cultured mammalian cells.
| Purity | : | 99.8 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 401 |
| ¡¡Route | : | Oral (gavage) |
| ¡¡Dosage | : | 0 (vehicle), 300, 500, 900, 1500 mg/kg |
| ¡¡Number of animals/group | : | Males, 5; females, 5 |
| ¡¡Vehicle | : | Corn oil |
| GLP | : | Yes |
¡¡Test results:
LD50: Males, 734 mg/kg; females, 671 mg/kg
| Purity | : | 99.8 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | Guideline for the 28-Day Repeated Dose Toxicity Test in Mammalian Species (Chemical Substances Control Law of Japan) |
| ¡¡Route | : | Oral (gavage) |
| ¡¡Dosage | : | 0 (vehicle), 10, 50, 250 mg/kg/day |
| ¡¡Number of animals/group | Males, 6; females, 6 | |
| ¡¡Vehicle | : | Corn oil |
| ¡¡Administration period | : | Males and females, 28 days |
| ¡¡Terminal killing | : | Days 29 or 43 |
| GLP | : | Yes |
¡¡Test results:
The NOEL is considered to be 10 mg/kg /day for both sexes.
| Purity | : | 99.8 % |
| Test species/strains | : | Salmonella typhimurium TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 471 |
| ¡¡Procedures | : | Pre-incubation method |
| ¡¡Solvent | : | Dimethyl sulfoxide |
| ¡¡Positive controls | : | -S9 mix; 2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide (TA100, TA98 and WP2 uvrA), Sodium azide (TA1535) and 2-Methoxy-6-chloro-9-[3-(2-chloroethyl)-aminopropylamino]acridine¡¦2HCl (TA1537) +S9 mix; Benzo[a]pyrene (TA100, TA98, TA1537) and 2-Aminoanthracene (TA1535 and WP2 uvrA) |
| ¡¡Dosage | : | -S9 mix; 0, 19.5, 39.1, 78.1, 156, 313, 625, 1250 ¦Ìg/plate (all strains) +S9 mix; 0, 19.5, 39.1, 78.1, 156, 313, 625, 1250 ¦Ìg/plate (all strains) |
| ¡¡S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ¡¡Plates/test | : | 3 |
| GLP | : | Yes |
¡¡Test results:
Genetic effects:
S. typhimurium TA100, TA1535
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [*] | [ ] | [ ] |
Genetic effects:
S. typhimurium TA98, TA1537
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
E. coli WP2 uvrA
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99.8 % |
| Type of cell used | : | Chinese hamster lung (CHL/IU) cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD test Guideline 473 |
| ¡¡Solvent | : | Dimethyl sulfoxide |
| ¡¡Positive control | : | -S9 mix; Mitomycin C +S9 mix; Cyclophosphamide |
| ¡¡Dosage | : | -S9 mix (short-term treatment); 0, 60, 80, 100, 120, 140, 160 and 180 ¦Ìg/mL +S9 mix (short-term treatment); 0, 60, 80, 100, 120, 140, 160 and 180 ¦Ìg/mL |
| ¡¡S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ¡¡Plates/test | : | 2 |
| GLP | : | Yes |
¡¡Test results:
| Lowest concentration producing cytogenetic effects in vitro | : | ||
| Without metabolic activation (short-term treatment) | : | 100 ¦Ìg/mL (clastogenicity) | |
| With metabolic activation (short-term treatment) | : | 60 ¦Ìg/mL (clastogenicity) | |
Genetic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| Without metabolic activation: | [*] | [ ] | [ ] | [ ] | [ ] | [*] |
| With metabolic activation: | [*] | [ ] | [ ] | [ ] | [ ] | [*] |
| 1) | The tests were performed by the Bozo Research Center Inc., 1284 Kamado, Gotemba-shi, Shizuoka, 412-0039, Japan. Tel +81-550-82-2000 Fax +81-550-82-2379 |