
In the single dose oral toxicity test, an LD50 value of more than 2000 mg/kg was found for both sexes.
With regard to repeated dose toxcity, no clinical signs were observed in males and females during the administration period. A tendency for suppression of body weight gain was detected in males given the 1000 mg/kg dose. On urinalysis, brown colored urine in males and females, an increase in specific gravity and decreases in pH and protein were detected in females at a dose of 1000 mg/kg. Hematological examination revealed prolonged prothrombin time in females at doses of 300 and 1000 mg/kg. Blood chemical examination revealed increases in triglyceride and total bilirubin in males, and increases in ¦Ć-GTP and total bilirubin, and decreases in potassium and chloride in females at a dose of 1000 mg/kg. There was a tendency for increase of absolute and relative thyroid weights in females, along with increases in absolute and relative liver weights in males and increase in the relative liver weight in females at a dose of 1000 mg/kg. Histopathological examination revealed hypertrophy of the thyroid follicular cell in both sexes at a dose of 1000 mg/kg. These changes attributable to administration of the test compound had disappeared at the termination of the recovery period. The NOEL is considered to be 300 mg/kg/day for both sexes.
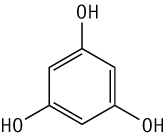
Genotoxicity of 1,3,5-trihydroxybenzene was studied by a reverse mutation test in bacteria. This substance was not mutagenic in Salmonella typhimurium TA100, TA1535, TA98, TA1537 or Escherichia coli WP2 uvrA, with or without an exogenous metabolic activation system.
Genotoxicity of 1,3,5-trihydroxybenzene was studied by a chromosomal aberration test in cultured Chinese hamster lung (CHL/IU) cells.
1,3,5-Trihydroxybenzene induced structural chromosomal aberrations under the present conditions with short-term treatment without metabolic activation and with continuous treatment. Furthermore, it induced polyploidy with the short-trem treatment without metabolic activation.
| Purity | : | 99.9 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 401 |
| ””Route | : | Oral (gavage) |
| ””Dosage | : | 0 (vehicle), 1000, 2000 mg/kg |
| ””Number of animals/group | : | Males, 5; females, 5 |
| ””Vehicle | : | Olive oil |
| GLP | : | Yes |
””Test results:
The LD50 value was estimated to be more than 2000 mg/kg for males and females.
| Purity | : | 99.9 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | Guideline for 28-Day Repeated Dose Toxicity Test in Mammalian Species (Chemical Substances Control Law of Japan) |
| ””Route | : | Oral (gavage) |
| ””Dosage | : | 0 (vehicle), 30, 100, 300, 1000 mg/kg |
| ””Number of animals/group | Males, 6; females, 6 | |
| ””Vehicle | Olive oil | |
| ””Administration period | : | Males and females, 28 days |
| ””Terminal killing | : | Males and females , days 29 or 43 |
| GLP | : | Yes |
””Test results:
| Purity | : | 100 wt% |
| Test species/strains | : | Salmonella typhimurium TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 471 |
| ””Procedures | : | Pre-incubation method |
| ””Solvent | : | Dimethyl sulfoxide |
| ””Positive controls | : | -S9 mix; 2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide (TA100, TA98, WP2 uvrA), Sodium azide (TA1535) and 9-Aminoacridine (TA1537) +S9 mix; 2-Aminoanthracene (five strains) |
| ””Dosage | : | -S9 mix; 0, 313, 625, 1250, 2500, 5000 ¦Ģg/plate (five strains) +S9 mix; 0, 313, 625, 1250, 2500, 5000 ¦Ģg/plate (five strains) |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 (1 for cytotoxicity test) |
| ””Number of replicates | : | 2 (plus 1 cytotoxicity test) |
| GLP | : | Yes |
””Test results:
Genetic effects:
Salmonella typhimurium TA100, TA98, TA1535, TA1537
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
Escherichia coli WP2 uvrA
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 100 wt% |
| Type of cell used | : | Chinese hamster lung (CHL/IU) cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 473 |
| ””Solvent | : | Distilled water for injection |
| ””Positive controls | : | -S9 mix; Mitomycin C +S9 mix; Cyclophosphamide |
| ””Dosage | : | -S9 mix (6 hr short-term treatment); 0, 0.33, 0.65, 1.3 mg/mL +S9 mix (6 hr short-term treatment); 0, 0.081, 0.16, 0.33 mg/mL -S9 mix (24 hr continuous treatment); 0, 0.081, 0.16, 0.33 mg/mL |
| ””S9 | : | Rat liver, induced with phenobarbital and 5, 6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
””Test results:
| Lowest concentration producing cytogenetic effects in vitro | : | |
| Without metabolic activation (short-term treatment) | : | 1.3 mg/mL (polyploidy) 1.3 mg/mL (clastogenicity) |
| Without metabolic activation (continuous treatment) | : | 0.16 mg/mL (clastogenicity) |
Genotoxic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| Without metabolic activation: | [*] | [ ] | [ ] | [*] | [ ] | [ ] |
| With metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| 1) | The tests were performed by the Research Institute for Animal Science in Biochemistry and Toxicology, 3-7-11 Hashimotodai, Sagamihara-shi, Kanagawa, 229-1132, Japan. Tel +81-42-762-2775 Fax +81-42-762-7979 |
| 2) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257-8523, Japan. Tel +81-463-82-4751 Fax +81-463-82-9627 |