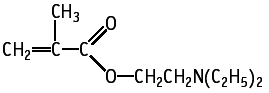

2-(Diethylamino)ethyl methacrylate was studied for oral toxicity in rats in an OECD combined repeat dose and reproductive/developmental toxicity screening test at doses of 0, 50, 150 and 500 mg/kg/day.
With regard to repeat dose toxicity, high values for BUN and relative weights of kidneys were observed in males of the 150 mg/kg group and high values for BUN and absolute and relative weights of kidneys were observed in males of the 500 mg/kg group. Low values for hemoglobin, mean corpuscular volume, mean corpuscular hemoglobin and mean corpuscular hemoglobin concentration and high values for total iron-binding capacity and unsaturated iron-binding capacity were seen, as well as low ¦Į2-globulin and high ¦Ā-globulin fraction ratios which were thought to be reflections of the aforementioned changes. There were no effects of the test substance in females of the 500 mg/kg group.
The NOELs for repeat dose toxicity are considered to be 50 mg/kg/day for males, and 500 mg/kg/day for females.
With regard to reproductive/developmental toxicity, a tendency toward low numbers of pups and low delivery index were observed in the 500 mg/kg group. There were no effects of the test substance on development of pups.
The NOELs for reproductive performance are considered to be 500 mg/kg/day for males, 150 mg/kg/day for females, and 500 mg/kg/day for development of pups.
2-(Diethylamino)ethyl methacrylate was not mutagenic in Salmonella typhimurium TA100, TA1535, TA98, TA1537 and Escherichia coli WP2 uvrA, with or without an exogenous metabolic activation system.
Genotoxicity of 2-(diethylamino)ethyl methacrylate was studied by the chromosomal aberration test in cultured Chinese hamster lung(CHL/IU)cells.
2-(Diethylamino)ethyl methacrylate induced structural chromosomal aberrations at 0.30 and 0.60 mg/mL(mid and high concentrations) in the presence of an exogenous metabolic activation system. Polyploidy was induced at 0.15 and 0.30 mg/mL(mid and high concentrations) on continuous treatment for 24 h, at 0.30 mg/mL(high concentration) on continuous treatment for 48 h and at 0.30 mg/mL(high concentration) in the absence of an exogenous metabolic activation system.
| Purity | : | 99.8 % |
| Test Species/Strain | : | Rat/Crj:CD(SD) |
| Test method | : | OECD Combined Repeat Dose and Reproductive/ Developmental Toxicity Screening Test |
| ””Route | : | Oral(gavage) |
| ””Dose | : | 0(Vehicle), 50, 150 and 500 mg/kg/day |
| ””Number of animals/group | : | Males, 12; females, 12 |
| ””Vehicle | : | Olive oil |
| ””Administration period | : | Males, 49 days Females, from 14 days before mating to the day before autopsy(day 3 of lactation) |
| ””Terminal sacrifice | : | Males, day 50 of administration Females, day 4 of lactation |
| GLP | : | Yes |
””Test results:
In males of the 500 mg/kg group, low values for hemoglobin, mean corpuscular volume, mean corpuscular hemoglobin and mean corpuscular hemoglobin concentration and high values for unsaturated iron-binding capacity and total iron-binding capacity were seen, as well as low ¦Į2-globulin and high ¦Ā-globulin fraction ratios, which were thought to be reflections of the aforementioned changes. In addition, high values for BUN and absolute and relative weights of kidneys were observed. There were no effects of the test substance on clinical signs, body weights, food consumption, urinalysis parameters, gross pathology or histopathology. In the 150 mg/kg group, high values for BUN and relative weights of kidneys were observed. There were no effects of the test substance on clinical signs, body weights, food consumption, urinalysis data, hematology, gross pathology or histopathology. In the 50 mg/kg group, there were no effects of the test substance with regard to any observation, measurement or examination. As for females, there were no effects of the test substance on clinical signs, body weights, food consumption or histopathology in any treatment groups.
The NOELs for repeat dose toxicity are considered to be 50 mg/kg/day for males, and 500 mg/kg/day for females.
<Reproductive/developmental toxicity>
With regard to male and female parents, there were no effects of the test substance on estrous cycle, copulation index, fertility index, length of gestation, delivery and lactation condition, the numbers of corpora lutea and implantation sites, implantation index or birth index. However, a tendency toward low numbers of pups born and a low delivery index were observed for the 500 mg/kg group.
With regard to pups, there were no effects of the test substance on sex ratio, the number of stillborn pups, live birth index, external anomalies, body weights, viability index on day 4 or gross pathology.
The NOELs for the reproductive/developmental toxicity are considered to be 500 mg/kg/day for males, 150 mg/kg/day for females, and 500 mg/kg/day for pups.
| Purity | : | 99.8 % |
| Test species/strains | : | Salmonella typhimurium TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA |
| Test method | : | Guidelines for Screening Toxicity Testing of Chemicals (Japan) and OECD Guidelines No. 471 and 472 |
| ””Procedures | : | Pre-incubation method |
| ””Solvent | : | DMSO |
| ””Positive controls | : | -S9 mix; 2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide(TA100, TA98, WP2 uvrA), Sodium azide(TA1535)and 9-Aminoacridine(TA1537) +S9 mix; 2-Aminoanthracene(five strains) |
| ””Doses | : | -S9 mix; 0, 156, 313, 625, 1250, 2500, 5000 ¦Ģg/plate(TA100, TA1535, TA98, TA1537); 0, 313 - 5000 ¦Ģg/plate(WP2 uvrA) +S9 mix; 0, 156 - 5000 ¦Ģg/plate(five strains) |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
””Test results:
Genetic effects:
Salmonella typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
Escherichia coli WP2 uvrA
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99.8 % |
| Type of cell used | : | Chinese hamster lung(CHL/IU)cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals(Japan) and OECD Guideline No. 473 |
| ””Solvent | : | DMSO |
| ””Positive controls | : | -S9 mix, Mitomycin C +S9 mix, Cyclophosphamide |
| ””Doses | : | -S9 mix(continuous treatment): 0, 0.075, 0.15, 0.30 mg/mL -S9 mix(short-term treatment): 0, 0.075, 0.15, 0.30 mg/mL +S9 mix(short-term treatment): 0, 0.15, 0.30, 0.60 mg/mL |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
””Test results:
Structural chromosomal aberrations(included gap) were induced at 0.30 and 0.60 mg/mL(mid and high concentrations, 6.0% and 24.5%, respectively) in the presence of an exogenous metabolic activation system. Polyploidy was induced under the following conditions: 24 h continuous treatment(0.15 and 0.30 mg/mL:mid and high concentration, 1.75 and 7.25%, respectively); 48 h continuous treatment(0.30 mg/mL, 5.13%); short-term treatment without an exogenous metabolic activation system(0.30 mg/mL, 7.13%).
Lowest concentration producing cytogenetic effects in vitro:
Without metabolic activation(continuous treatment ): 0.15 mg/mL(polyploidy)
Without metabolic activation(short-term treatment): 0.30 mg/mL(polyploidy)
With metabolic activation(short-term treatment): 0.30 mg/mL(clastogenicity)
Genotoxic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] | [*] | [ ] | [ ] |
| With metabolic activation: | [*] | [ ] | [ ] | [ ] | [ ] | [*] |
| 1) | The tests were performed by Bozo Research Center Inc, 1284, Kamado, Gotemba-shi, Shizuoka, 412-0039, Japan. Tel +81-550-82-2000 Fax +81-550-82-2379 |
| 2) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257-0025, Japan. Tel +81-463-82-4751 Fax +81-463-82-9627 |