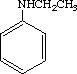

The single dose toxicity test revealed LD50 values of 382 mg/kg for males and of 553 mg/kg for females.
¡¡In the repeat dose toxicity test, hemolytic anemia accompanied by methemoglobinemia and formation of erythrocytic Heinz bodies, shortening of prothrombin time and increases of liver and spleen weights were observed in both sexes in a dose-related fashion. Histopathological examination revealed hemosiderosis of the spleen, liver and kidney, increased erythropoiesis in the bone marrow, spleen and liver, and decreased lymphocytes in the marginal zone of the splenic lymph follicles both sexes at a doses of 5 mg/kg and above demonstrated a tendency for disappearance. All changes related to the test substance disappeared or after a 14-day recovery period. The NOELs are considered to be 1 mg/kg/day for both sexes.
¡¡N-Ethylaniline was not mutagenic in Salmonella typhimurium TA100, TA98, TA1535, TA1537 and Escherichia coli WP2 uvrA.
¡¡This chemical substance induced structural chromosomal aberrations in CHL/IU cells at the highest concentration (10 mM = 1.1 mg/ml) after 6-hr treatment without exogenous metabolic activation. Polyploid cells were not induced under the present experimental conditions.
| Purity | : | 99.6% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | OECD Guideline 401 |
| ¡¡Dosage | : | 0 (vehicle), 240, 300, 375, 469, 586, 732 mg/kg |
| ¡¡Number of animals | : | Males, 5; females, 5/group |
| ¡¡GLP | : | Yes |
| Purity | : | 99.6% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | Guideline for 28-day Repeat Dose Toxicity Testing of Chemicals (Japan) |
| ¡¡Route | : | Oral (gavage) |
| ¡¡Dosage | : | 0 (vehicle), 1, 5, 25, 125 mg/kg/day |
| ¡¡Number of animals | : | Males, 6; females, 6/group |
| ¡¡Vehicle | : | Sesame oil |
| ¡¡Administration period | : | Males and females, 28 days |
| ¡¡Terminal kill | : | Males and females, day 29 or 43 |
| ¡¡GLP | : | Yes |
| Purity | : | 99.6% |
| Test species/strains | : | S.typhimurium TA100, TAl535, TA98, TA1537 |
| E. coli WP2 uvrA | ||
| Test methods | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ¡¡Procedures | : | Plate incorporation method |
| ¡¡Solvent | : | DMSO |
| ¡¡Positive controls | : | -S9 Mix, AF-2 (TA100, WP2, TA98), sodium azide (TA1535) and 9-aminoacridine (TA1537) +S9 Mix, 2-aminoanthracene (all strains) |
| ¡¡Doses | : | 0, 78.13, 156.3, 312.5, 625, 1250 and 2500 ¦Ìg/plate |
| ¡¡S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ¡¡Plates/test | : | 3 |
| ¡¡Number of replicates | : | 2 |
| GLP | : | Yes |
| + | ? | - | |
| without metabolic activation: | [ ] | [ ] | [*] |
| with metabolic activation: | [ ] | [ ] | [*] |
| + | ? | - | |
| without metabolic activation: | [ ] | [ ] | [*] |
| with metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99.6% |
| Type of cell used | : | Chinese hamster lung (CHL/IU) cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ¡¡Solvent | : | Dimethyl sulfoxide |
| ¡¡Positive controls | : | -S9, Mitomycin C +S9, Cyclophosphamide |
| ¡¡Doses | : | -S9 (continuous treatment):0, 0.3, 0.6, 1.1 mg/ml -S9 (short-term treatment):0, 0.3, 0.6, 1.1 mg/ml +S9 (short-term treatment):0, 0.3, 0.6, 1.1 mg/ml |
| ¡¡S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ¡¡Plates/test | : | 2 |
| GLP | : | Yes |
| without metabolic activation (continuous treatment ) | : | 0.6 mg/ml (clastogenicity) |
| without metabolic activation (short-term treatment) | : | 1.1 mg/ml (clastogenicity) |
¡¡Genotoxic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| without metabolic activation: | [*] | [ ] | [ ] | [ ] | [ ] | [*] |
| with metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| 1) | The tests were performed by the Research Institute for Animal Science in Biochemistry and Toxicology, 3-7-11 Hashimotodai, Sagamihara-shi, Kanagawa 229, Japan.¡¡Tel¡¡+81-427-62-2775¡¡Fax¡¡+81-427-62-7979 |
| 2) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257, Japan.¡¡Tel¡¡+81-463-82-4751¡¡Fax¡¡+81-463-82-9627 |