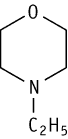

4-Ethylmorpholine was studied in rats of both sexes with the guideline for repeated dose 28-day oral toxicity study in rodents (OECD Test Guideline 407) at doses of 0, 50, 200 and 800 mg/kg.
No deaths were observed in any treatment group. Clinical signs of intermittent cage-licking and chewing were observed in the groups of 200 and 800 mg/kg. In each sex receiving 800 mg/kg, action tremors, decrease in movement, a crouching position and eyelid closure were observed. Depressed body weight gain was noted with decrease of food consumption in each sex. Blood biochemical findings revealed increases in inorganic phosphate and decreases in chloride. Pathological findings were as follows: increase in liver weights and kidney weights, hypertrophy of hepatocyte in the centrilobular, vacuolation of epithelium in distal and Henle's loop. From the results, the NOELs are considered to be 50 mg/kg/day for males and females.
4-Ethylmorpholine was evaluated for reproductive/developmental toxicity in Sprague-Dawley rats according to the OECD Test Guideline 421 at doses of 0, 50, 150 and 500 mg/kg. Male rats were orally administered for 42 days from 2 weeks before mating to the necropsy, and female rats were orally administered from 2 weeks before mating through gestation until day 3 of lactation.
Although one female in the 500 mg/kg group died on day 2 of lactation, no deaths were found in any groups of males. In general conditions, tremors were observed in the dead female, and temporary salivation after dosing was observed in males and females of the 150 mg/kg or more groups. Furthermore, decreases in body weight gain and food consumption were detected in males of the 500 mg/kg group and females of the 150 mg/kg or more groups. Necropsy and histopathological examination of reproductive organs revealed no abnormalities related to dosing.
Slight decreases in numbers and rate of implantation were detected in the 500 mg/kg group. There were, however, no adverse effects on the estrous cycle, copulation, fertility, gestation length, gestation index or number of corpora lutea. Slight decreases in numbers of pups and numbers of live pups, possibly related to the decrease in implantation, were detected in the 500 mg/kg group. There were no changes related to dosing in morphology or body weights of pups.
In conclusion, the NOELs for parent animals are considered to be 150 mg/kg/day for males and 50 mg/kg/day for females, because decrease in body weight gain was detected. The NOEL for reproduction is considered to be 150 mg/kg/day for parent animals, and the NOEL for offspring is considered to be 500 mg/kg/day.
Genotoxicity of 4-ethylmorpholine was studied by a reverse mutation test in bacteria and by a chromosomal aberration test in cultured Chinese hamster lung (CHL/IU) cells.
4-Ethylmorpholine was not mutagenic in Salmonella typhimurium TA100, TA1535, TA98, TA1537 or Escherichia coli WP2 uvrA, with or without an exogenous metabolic activation system.
4-Ethylmorpholine did not induce chromosomal aberrations in CHL/IU cells, with or without an exogenous metabolic activation system.
| Purity | : | ”ę99 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 401 |
| ””Route | : | Oral (Gavage) |
| ””Dosage | : | 0 (Vehicle), 500, 1000, 1500, 2000 mg/kg |
| ””Number of animals/group | : | Males, 5; females, 5 |
| ””Vehicle | : | Water for injection |
| GLP | : | Yes |
””Test results:
| Purity | : | ”ę99 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 407 |
| ””Route | : | Oral (Gavage) |
| ””Dosage | : | 0 (Vehicle), 50, 200, 800 mg/kg/day |
| ””Number of animals/group | : | Males, 10; females, 10 (0, 800 mg/kg) Males, 5; females, 5 (50, 200 mg/kg) |
| ””Vehicle | : | Water for injection |
| ””Administration period | : | Males and females, 28 days |
| ””Terminal killing | : | Males and females, on days 29 and 43 |
| GLP | : | Yes |
””Test results:
From the results, the NOEL is considered to be 50 mg/kg/day for males and females.
| Purity | : | ”ę99 % |
| Test species/strain | : | Rat/Crj:CD(SD)IGS |
| Test method | : | OECD Test Guideline 421 |
| ””Route | : | Oral (Gavage) |
| ””Dosage | : | 0 (Vehicle), 50, 150, 500 mg/kg/day |
| ””Number of animals/group | : | Males, 13; females, 13 |
| ””Vehicle | : | Water for injection |
| ””Administration period | : | Males, 42 days Females, from 14 days before mating to day 3 of lactation |
| ””Terminal killing | : | Males, day 43 of administration Females, day 4 of lactation |
| GLP | : | Yes |
””Test results:
| Purity | : | 99.84 % |
| Test species/strain | : | Salmonella typhimurium TA100, TA1535, TA98, TA1537, Escherichia coli WP2 uvrA |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 471 |
| ””Procedures | : | Pre-incubation method |
| ””Solvent | : | Water for injection |
| ””Positive controls | : | -S9 mix; 2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide (TA100, TA98, WP2 uvrA), Sodium azide (TA1535) and 9-Amino-acridine (TA1537) +S9 mix; 2-Aminoanthracene (all strains) |
| ””Dosage | : | -S9 mix ; 0, 313, 625, 1250, 2500, 5000 ¦Ģg/plate +S9 mix ; 0, 313, 625, 1250, 2500, 5000 ¦Ģg/plate |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
””Test results:
Genotoxic effects:
Salmonella typhimurium TA100, TA1535, TA98, TA1537
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
Escherichia coli WP2 uvrA
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99.84 % |
| Type of cell used | : | Chinese hamster lung(CHL/IU)cells |
| Test method | : | Guidelines for Screening Mutagenicity Testing of Chemicals (Chemical Substances Control Law of Japan) and OECD Test Guideline 473 |
| ””Solvent | : | Isotonic sodium chloride solution |
| ””Positive controls | : | -S9 mix; 1-Methyl-3-nitro-1-nitrosoguanidine +S9 mix; 3,4-Benzo[a]pyrene |
| ””Dosage | : | -S9 mix (6 hr short-term treatment); 0, 150, 300, 600, 1200 ¦Ģg/mL +S9 mix (6 hr short-term treatment); 0, 150, 300, 600, 1200 ¦Ģg/mL -S9 mix (24 hr continuous treatment); 0, 150, 300, 600, 1200 ¦Ģg/mL |
| ””S9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plates/test | : | 2 |
| GLP | : | Yes |
””Test results:
Cytotoxicity was observed at 1200 ¦Ģg/mL after 24 hr continuous treatment without S9 mix.
Genotoxic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| 1) | The tests were performed by the Hatano Research Institute, Food and Drug Safety Center, 729-5 Ochiai, Hadano-shi, Kanagawa, 257-8523, Japan. Tel +81-463-82-4751””Fax +81-463-82-9627 |
| 2) | The tests were performed by the Research Institute for Animal Science in Biochemistry and Toxicology, 3-7-11 Hashimotodai, Sagamihara-shi, Kanagawa, 229-1132, Japan. Tel +81-42-762-2775””Fax +81-42-762-7979 |