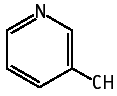

3-Cyanopyridine was studied for oral toxicity in rats of both sexes in a single dose toxicity test at doses of 0, 1297, 1388, 1485, 1589 and 1700 mg/kg. This revealed LD50 values of 1475 mg/kg for males and 1455 mg/kg for females.
The compound was then studied for oral toxicity in rats in a 28-day repeat dose toxicity test at doses of 0, 5, 30 and 180 mg/kg.
Salivation was observed in males and females of the 180 mg/kg group just after administration from the mid stage of the administration period, and lacrimation was infrequently observed in 1 female of the 180 mg/kg group about 6 hours after administration. Body weight gain of males of the 180 mg/kg group was suppressed during the administration period. Food consumption decreased in males and females of the 180 mg/kg group during the early administration period. Urinalysis showed increase in urine volume, decreases in osmotic pressure, specific gravity and pH, and pale coloration in males and females of the 180 mg/kg group. Hematological examination showed increases in leukocyte counts, the reticulocyte ratio, MCV, MCH and decrease in erythrocyte counts in males and females of the 180 mg/kg group, and increase in the segmented neutrophil ratio and decrease in the lymphocyte ratio in males of the 180 mg/kg group. Blood chemical examination showed increases in total protein, albumin, the A/G ratio, GPT, total cholesterol and phospholipids in males and females of the 180 mg/kg group, and decrease in triglycerides in males and decreases in cholinesterase and acetylcholinesterase in females of the 180 mg/kg group. Liver and kidney weights were increased in males and females of the 30 mg/kg group, and along with adrenal weights in the 180 mg/kg group. Histopathologically, centrilobular hypertrophy of hepatocytes was observed in males and females of the 30 mg/kg and higher dose groups. The incidence of hyaline droplets in proximal tubules was also increased in males of the 30 mg/kg and higher dosage groups. Furthermore, hypertrophy of zona fasciculata of the adrenals and extramedullary hematopoiesis and hemosiderin deposits in the spleen were observed in males and females of the 180 mg/kg group. Necrosis of spermatocytes and round type spermatids as well as decreased elongate type spermatids and vacuolation of Sertoli cells were evident in males of the 180 mg/kg group. In addition, cystitis and neutrophilic infiltration in the renal pelvis were observed in 1 female of the 180 mg/kg group. In the recovery test, all changes observed during the administration period demonstrated recovery. The NOEL is considered to be 5 mg/kg/day for both sexes under the conditions of the present study.
3-Cyanopyridine was not mutagenic in Salmonella typhimurium, TA100, TA1535, TA98, TA1537 and Escherichia coli WP2 uvrA. Regardless of the presence or absence of exogenous metabolic activation system, this chemical substance did not induce structural chromosomal aberrations in CHL/IU cells. Polyploidy was not observed under the conditions of the present study.
| Purity | : | 99.9% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | OECD Test Guideline 401 |
| ””Route | : | Oral (gavage) |
| ””Doses | : | 0 (Vehicle), 1297, 1388, 1485, 1589, 1700 mg/kg/day |
| ””Number of animals/group | : | Males, 5; females, 5 |
| ””Vehicle | : | Water for injection |
| GLP | : | Yes |
””Test results:
The LD50 values were 1475 mg/kg for males and 1455 mg/kg for females.
| Purity | : | 99.9% |
| Test species/strain | : | Rat/Crj:CD (SD) |
| Test method | : | Guidelines for 28-day Repeat Dose Toxicity Testing of Chemicals (Japan) |
| ””Route | : | Oral (gavage) |
| ””Dosage | : | 0 (Vehicle), 5, 30, 180 mg/kg/day |
| ””Number of animals | : | Males, 6 or 12 (0, 180 mg/kg) Females, 6 or 12 (0, 180 mg/kg) |
| ””Vehicle | : | Water for injection |
| ””Administration period | : | Males and females, 28 days |
| ””Terminal kill | : | Males and females, days 29 or 43 (0, 180 mg/kg) |
| GLP | : | Yes |
””Test results:
The NOEL was considered to be 5 mg/kg/day for both sexes under the conditions of the present study.
| Purity | : | 99.9 % |
| Test species/strain | : | Salmonella typhimurium, TA100, TA1535, TA98, TA1537 Escherichia coli WP2 uvrA |
| Test method | : | OECD Guidelines No. 471, 472 and Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ””Procedures | : | Pre-incubation method |
| ””Solvent | : | DW |
| ””Positive controls | : | -S9 mix, AF-2 (TA100, TA98), Sodium azide (TA1535), ENNG (WP2 uvrA) and 9-Aminoacridine (TA1537) +S9 mix, 2-Aminoanthracene (all strains) |
| ””Doses | : | -S9 mix; 313, 625, 1250, 2560, 5000 ¦Ģg/plate +S9 mix; 313, 625, 1250 ,2560, 5000 ¦Ģg/plate |
| ””S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plate/test | : | 3 |
| ””Number of replicates | : | 2 |
| GLP | : | Yes |
””Test results:
Genotoxic effects:
S. typhimurium, TA100, TA1535, TA98, TA1537
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
E. coli WP2 uvrA
| + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] |
| Purity | : | 99.9 % |
| Type of cell used | : | Chinese hamster CHL/IU cells |
| Test method | : | OECD Guidelines No. 473 and Guidelines for Screening Mutagenicity Testing of Chemicals (Japan) |
| ””Solvent | : | JP saline |
| ””Positive controls | : | -S9 mix, Mitomycin C +S9 mix, Benzo[a]pyrene |
| ””Doses | : | -S9 mix (24 hr continuous treatment): 0, 375, 750, 1500, 3000 ¦Ģg/ml -S9 mix (48 hr continuous treatment): 0, 375, 750, 1500, 3000 ¦Ģg/ml -S9 mix (6 hr short-term treatment): 0, 625, 1250, 2500, 5000 ¦Ģg/ml +S9 mix (6 hr short-term treatment): 0, 1250, 2500, 5000 ¦Ģg/ml |
| ””S-9 | : | Rat liver, induced with phenobarbital and 5,6-benzoflavone |
| ””Plate/test | : | 2 |
| GLP | : | Yes |
””Test results:
Genotoxic effects:
| clastogenicity | polyploidy | |||||
| + | ? | - | + | ? | - | |
| Without metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| With metabolic activation: | [ ] | [ ] | [*] | [ ] | [ ] | [*] |
| 1) | The tests were performed by Safety Assessment Laboratory, Panapharm Laboratories Co., Ltd. 1285 Kurisaki-machi, Uto-shi, Kumamoto, 869-04, Japan Tel +81-964-23-5111 Fax +81-964-23-2282 |
| 2) | The tests were performed by the Mitsubishi Chemical Safety Institute Ltd., 14 Sunayama, Hasaki-machi, Kashima-gun, Ibaraki, 314-02, Japan. Tel +81-479-46-2871 Fax +81-479-46-2874 |