
 |
|
Printing
Date |
2017-01-04 16:44:19
JST |
Restriction
of specific regulatory purposes | |
Confidentiality
| |
|
Name
|
chlorocyclohexane
|
|
Legal
entity owner |
National Institute of
Health Sciences / Tokyo / Japan |
|
UUID
|
IUC5-cd10453c-72f2-45d4-a87e-fc2882f22d3b
| |
|
Dossier
UUID |
0
| |
|
Author
|
dra /
National Institute of Health Sciences / Tokyo / Japan | |
|
Date
|
2017-01-04
16:41:51 JST | |
|
Remarks
|
|
Chemical
name |
chlorocyclohexane
|
|
Legal
entity |
|
|||||||||||||
|
UUID
|
IUC5-6f52b438-c5b8-41b0-b587-ae524ef28122
| |
|
Dossier
UUID |
0
| |
|
Author
|
dra /
National Institute of Health Sciences / Tokyo / Japan | |
|
Date
|
2017-01-04
16:42:38 JST | |
|
Remarks
|
|
Purpose
flag |
key study
| ||
|
Study
result type |
experimental result
|
||
|
Reliability
|
1
(reliable without restriction) | ||
|
Rationale
for reliability incl. deficiencies |
OECD Test
Guideline study under GLP condition | ||
|
Reference
type |
Author
|
Year
|
Title
|
Bibliographic
source |
Testing
laboratory |
Report
no. |
Owner
company |
Company
study no. |
Report
date |
|
study report |
MHLW (Ministry of Health, Labour and Welfare), Japan |
2007 |
Single Dose Oral Toxicity Test of Chlorocyclohexane in Rats |
Japan Existing Chemical Data Base (JECDB) |
Mitsubishi Safety Institute Ltd. |
B041793 |
2007-01-23 |
data published |
standard acute method |
yes |
|
Qualifier
|
Guideline
|
Deviations
|
|
according to |
OECD Guideline 423 (Acute Oral toxicity - Acute Toxic Class Method)
|
no |
yes |
yes |
|
Identifier
|
Identity
|
|
CAS number |
542-18-7 |
- Name of test material (as cited in study report):
Chlorocyclohexane - CAS No.: 542-18-7 - Molecular weight: 118.61 - Lot No.: 5C1114 - Purity: 99.7% - Supplier: Junsei Chemical Co., Ltd. - Vapor pressure: 6.73 mmHg (25°C) - Vapor density : 4.12 (air = 1) - Boiling point : 142°C - Melting point: -44°C - Flash point: °C - Specific gravity: 1.004 (20/4°C) - Solubility: Insoluble in water and miscible by organic solvents as the alcohol and ether et al. - Odor: Characteristic odor - Physical state: Slightly pale yellow from colorless liquid - Storage condition of test material: Dark and sealed in refrigerator (2.8 – 8.4°C) |
rat |
Crj: CD(SD) |
female |
oral: gavage |
olive oil |
VEHICLE- Concentration in vehicle: 60 and 400
mg/ml.MAXIMUM DOSE VOLUME APPLIED: 5 ml/kg b.w.
|
300 mg/kg bw (first and second administration) 2000 mg/kg bw (third and fourth administration) |
First and second administration (first purchase): 3
animals and 3 animals (animal ID No. 50101 – 50103 and 60101 - 60103)
/female/300 mg/kg dose Third and fourth administration (second purchase): 3 animals and 3 animals (animal ID No. 70101 – 70103 and 80101 - 80103) /female/2000 mg/kg dose |
no |
- Duration of observation period following
administration: 14 days - Frequency of observations: Before dosing, Day 1 (day of administration):10, and 30 minutes and 1, 3, and 6 hrs after administration. After day 2: once a day - Frequency of weighing: Days 1 (before administration), 4, 8 and 15 - Necropsy of survivors performed: Yes |
No |
Dose levels were determined by the Globally Harmonized
Classification System (GHS) (mg/kg b.w.). Mortality was not observed in
each first and second administrations of 300 mg/kg groups and in each
third and fourth administrations of 2000 mg/kg groups.
|
|
Sex
|
Endpoint
|
Effect
level |
Based on
|
95%
CL |
Remarks
|
|
female |
LD50 |
> 2000 |
No deaths were observed from first to fourth
administration. |
No changes related to the test substance were observed
from first to forth administration. |
No changes related to the test substance were observed
in first and second administrations of 300 mg/kg groups. Depression of
body weight gains or decreases in body weight was observed in third and
fourth administrations of 2000 mg/kg groups. However, their body weights
were recovered at Day 8 and were equivalent to 300 mg/kg groups at Day 15.
|
No changes related to the test substance were observed
from first to fourth administration. |
- Organ weights: No data- Histopathology: No data-
Potential target organs: Not identified- Other observations: No data
|
The LD50 value was more than 2000 mg/kg bw for
females. GHS classification was classified in Category 5 (>2000 – 5000 mg/kg b.w.). |
|
UUID
|
IUC5-32931199-101f-41ea-aa8c-9e2425253f75
| |
|
Dossier
UUID |
0
| |
|
Author
|
dra /
National Institute of Health Sciences / Tokyo / Japan | |
|
Date
|
2017-01-04
16:42:46 JST | |
|
Remarks
|
|
Purpose
flag |
key study
| ||
|
Study
result type |
experimental result
|
||
|
Reliability
|
1
(reliable without restriction) | ||
|
Rationale
for reliability incl. deficiencies |
OECD Test
Guideline study under GLP condition | ||
|
Reference
type |
Author
|
Year
|
Title
|
Bibliographic
source |
Testing
laboratory |
Report
no. |
Owner
company |
Company
study no. |
Report
date |
|
study report |
MHLW (Ministry of Health, Labour and Welfare), Japan |
2007 |
A combined repeated-dose/reproductive-developmental toxicity study of
chlorocyclohexane by oral administration in rats. |
Japan Existing Chemical Data Base (JECDB) |
Mitsubishi Safety Institute Ltd. |
B041794 |
2007-01-23 |
data published |
7.8.1 Toxicity to reproduction: Toxicity to
reproduction.001 |
combined repeated dose and reproduction / developmental
screening |
no |
|
Qualifier
|
Guideline
|
Deviations
|
|
according to |
OECD Guideline 422 (Combined Repeated Dose Toxicity Study with the
Reproduction / Developmental Toxicity Screening Test) |
no |
yes |
yes |
- Name of test material (as cited in study report):
Chlorocyclohexane - CAS No.: 542-18-7 - Molecular weight: 118.60 - Lot No.: 5C1114 - Purity: 99.7% - Supplier: Junsei Chemical Co., Ltd. - Vapor pressure: 6.73 mmHg (25°C) - Vapor density : 4.12 (air = 1) - Boiling point : 142°C - Melting point: -44°C - Flash point: 29°C - Specific gravity: 1.004 (20/4°C) - Solubility: Insoluble in water and miscible with organic solvents such as alcohol and ether etc. - Odor: Peculiar odor - Physical state: Colorless liquid to slightly pale yellow - Storage condition of test material: Dark and sealed in refrigerator (2.8 – 8.4°C) |
rat |
Crj: CD(SD) |
male/female |
TEST ANIMALS - Source: Atsugi Breeding Center, Charles River Laboratories Japan, Inc. - Age at study initiation: 9 weeks of age - Weight at study initiation: 325-386 g for males and 193-231 g for females - Housing: bracket-type metallic wire-mesh cages/males and females excluding gestation and lactation periods (W 195 × D 325 × H 180 mm), and polycarbonate cage during gestation and lactation periods/females (W 265 × D 426 × H 200 mm) - Water (e.g. ad libitum):ad libitum - Acclimation period: 5 days ENVIRONMENTAL CONDITIONS - Temperature (°C): 21.4 to 23.7°C - Humidity (%): 48.8 to 65.0% - Air changes (per hr): 6 to 20 times per hour - Photoperiod (hrs dark / hrs light):12-hour lighting per day |
oral: gavage |
olive oil |
PREPARATION OF DOSING SOLUTIONS: Test substance was
dissolved in olive oil for injection. VEHICLE - Justification for use and choice of vehicle: No data - Amount of vehicle (if gavage): 5 ml/kg bw - Lot/batch no. (if required): No data - Dosing volume: 5 mL/kg - Stability (test solutions): For 8 days - Storage condition of test solution: Stored in a dark place at room temperature |
yes |
Test suspensions at each concentration of initial and
final preparations were analyzed by the GC method at Mitsubishi Safety
Institute Ltd. Results showed that the concentration of the test article
in each suspension was 95.0 to 106.2% of the nominal concentration and
both values were within the acceptable range (concentration: percentage of
the nominal concentration, 100 ± 10%) |
(P) Males: 42 days including 14 days pre-mating and
mating periods (P) Females: Days including 14 days pre-mating, mating and gestation periods and the days until day 4 of lactation |
Daily: 7 times / week |
0 (vehicle), 10, 60 and 300 mg/kg bw/day | |
Basis
|
actual ingested |
12 females/dose (0, 10, 60, 300 mg/kg), 7, 12, 12, 7
males of 0, 10, 60, 300 mg/kg, respectively, 5 males and 5 females at 0
and 300 mg/kg bw/day (recovery group) |
yes, concurrent vehicle |
- Dose selection rationale: A preliminary study was
conducted to determine the doses to be employed. Three males and three
female SD rats receiving 0, 30, 100, 300, and 1000 mg/kg groups of the
substance were administered for 14 days. As a result, death or moribundity
was observed in all males and females receiving 1000 mg/kg groups.
Salivation and increases in absolute and relative adrenal weights were
observed in females receiving 300 mg/kg group. No clear changes related to
the test substance were observed in males receiving 300 mg/kg group.
Therefore, the high dose was set at 300 mg/kg/day, and the middle and low
dose were set at 60 and 10 mg/kg/day using common ratio 5. Vehicle control
groups were set using olive oil only. |
CAGE SIDE OBSERVATIONS: Yes - Time schedule: Males and females: once before the start of administration, during the administration period, and during the recovery period DETAILED CLINICAL OBSERVATIONS: Yes - Time schedule: Males: once before the start of administration, once every week until Week 6 during the administration Females: once before the start of administration, once every week until Week 6 during the administration and once during the lactation period. No observation performed during the recovery period because no observed clinical changes during the administration period. BODY WEIGHT: Yes - Time schedule for examinations: Males in the main and recovery groups were weighed on Day 1, 8, 15, 22, 29, 36, 42, and 43 of administration, and males of recovery groups were weighed on Day 50 and 56. Female satellite groups were weighted same frequencies to male recovery groups. Females in the main groups were weighed on Day 1, 8 and 15 of administration and copulated females were weighed on Day 0, 7, 14 and 20 of gestation, and days 0 and 4 of lactation. FOOD CONSUMPTION : Yes - Food consumption (g/day/rat) for each animal determined from the difference of the of the previous day's feeding amount: Yes Measurement of food consumption was conducted on all animals at the following frequencies: Males in the main and recovery groups on Day 1-8, 8-15, 15-22, 22-29, 29-36, 36-38, 43-50, and 50-52. Female satellite groups on Day 1-8, 8-15, 15-22, 22-29, 29-36, 36-42, 43-50, and 50-56. Females in the main group on Day 1-8, 8-15, 22-29, 29-36, 36-38, 43-50, and 50-52. FOOD INTAKE: No HAEMATOLOGY: Yes - Time schedule for collection of blood: On the day after the final day of administration and on the final day of the recovery period - Anaesthetic used for blood collection: Yes - Animals fasted: Yes - How many animals: 5 animals/sex/group - Parameters examined red blood cell count, hemoglobin, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, reticulocyte count, platelet count, white blood cell count, differential white blood cell count, prothrombin time, activated partial thromboplastin time CLINICAL CHEMISTRY: Yes - Time schedule for collection of blood: On the day after the final day of administration and on the ending day of the recovery period - Animals fasted: Yes - How many animals: 5 animals/sex/group - Parameters checked: ASAT (GOT), ALAT (GPT), γ-GT, ALP, total bilirubin, blood urea nitrogen, creatinine, glucose, total cholesterol, triglyceride, total protein, albumin, A/G ratio, calcium, inorganic phosphorus, sodium, potassium, chloride URINALYSIS: Yes - Time schedule for collection of urine: final week of administration (Day 38 of administration) in males - Metabolism cages used for collection of urine: Yes - Animals fasted: Yes - How many animals: 5 animals/males/group - Parameters checked: pH, protein, glucose, ketones, bilirubin, occult blood, urobilinogen BLOOD HORMONE: No NEUROBEHAVIOURAL EXAMINATION: Yes - Battery of functions tested: 1) Open field observation: Aerial righting reaction, arousal, urination, defecation, posture and body position, breathing, co-ordination movement, gait, tremor, clonic convulsion, tonic convulsion, stereotypy, and bizarre behavior. 2) Manipulative Test: Approach response, touch response, auditory response, tail pinch response, and aerial righting reflex 3) Measurement of Grip Strength: Grip strength of forelimb and hind limb 4) Measurement of Motor Activity.Motor activity: 10-minute intervals from 0 to 60 minutes |
GROSS PATHOLOGY AND ORGAN WEIGHTS:Brain, heart, liver,
kidneys, adrenals, thymus, spleen, testes, and
epididymis. HISTOPATHOLOGY: Cerebrum, pituitary, thymus, lymph nodes (including mesenteric and mandibular lymph nodes), trachea, lung, stomach, intestinal tract (duodenum, jejunum, ileum, cecum, colon, rectum), thyroids, parathyroid, heart, liver, spleen, kidneys, adrenals, urinary bladder, testes, epididymis, seminal vesicles (including the coagulating gland), prostate (ventral lobe), ovaries, uterus, vagina, bone marrow (one side femur), Sciatic nerve (one side femur), spinal cord, and gross abnormal sites. |
The data were analyzed for homogeneity of variance by
the Bartlett test. If variances were homogeneous, data was analyzed by
one-way analysis of variance, whereas heterogenous data was analyzed by
Kruskal-Wallis ranking test. When a significant difference was observed,
Dunnett’s multiple comparison test was conducted between control and
treated groups. If not homogenous, analysis was performed using the
Kruskal-Wallis ranking test. Qualitative value as the pathological
findings was analyzed by Wilcoxon test and Fisher’s exact test. Urinalyses
data were analyzed by Kruskal-Wallis and Dunnet’s type mean rank test.
Statistical significance was set at < 5% by two-sided.
|
|
Endpoint
|
Effect
level |
Based on
|
Sex
|
Basis
for effect level / Remarks |
|
NOAEL |
10 mg/kg bw/day (actual dose received) |
male |
Effects of lesions in the hyperplasia of mucosal epithelium of
urinary bladder in males receiving 60 mg/kg . | |
|
NOAEL |
60 mg/kg bw/day (actual dose received) |
female |
Effects of lesions in the hyperplasia of mucosal epithelium of
urinary bladder in females receiving 300 mg/kg.
|
yes |
yes |
no effects |
not examined |
no effects (Significant increase in reticulocyte counts
was observed in males receiving 300 mg/kg bw/day, but it was not
considered to be toxicological effects.) |
yes (Significant decrease in inorganic phosphorus
concentration was observed in females receiving 300 mg/kg bw/day, but it
was not considered to be toxicological effects.)
|
no effects |
no effects |
yes |
yes |
yes |
no data |
CLINICAL SIGNS AND MORTALITY: No animal died in any
group. Salivation was observed in males and females receiving 300 mg/kg
from Day 9 to the end of dosing. No abnormal changes were observed in both
sexes with recovery animals during the recovery period. DETAILED CLINICAL OBSERVATIONS, MANIPULATIVE TEST, GRIP STRENGTH TEST AND LOCOMOTOR ACTIVITY MEASUREMENT: There were no changes related to the test substance in any group during the dosing. BODY WEIGHT: Depression of body weight gains was observed in males and females receiving 300 mg/kg from Day 8 to 42. ORGAN WEIGHTS: A significant increase in relative kidney weight was observed in males receiving 300 mg/kg bw/day at the end of both administration and recovery periods. HISTOPATHOLOGY: Hyaline droplet of proximal tubular epithelium in kidneys was observed in all males receiving 300 mg/kg. Minimal hyperplasia of mucosal epithelium in urinary bladder was observed in males receiving 60 mg/kg and males and females receiving 300 mg/kg at the end of the administration. This pathological change was found in females receiving 300 mg/kg after the recovery period. Minimal cell infiltration was present in one male and one female receiving 300 mg/kg at the end of administration and recovery periods. |
Figures and Tables (in English) are available in the following full report of the study. http://dra4.nihs.go.jp/mhlw_data/home/pdf/PDF542-18-7d.pdf |
Based on the effects of chlorocyclohexane on the urinary
bladder, the no observed adverse effect level (NOAEL) for repeated oral
dosing was determined to be 10 mg/kg bw/day in male rats and 60 mg/kg
bw/day in female rats. |
A combined repeated oral dose toxicity study and reproduction/developmental toxicity screening test was performed according to the OECD TG 422. Male and female rats (12 animals/sex/dose) were administered chlorocyclohexane at 0, 10, 60, and 300 mg/kg bw/day. Males were dosed for 42 days, including a 14 day pre-mating period and subsequent mating period. Females were dosed for up to 55 days, including 14 day pre-mating, mating, and gestation periods, and the time until lactation day 4. Five out of 12 males dosed at 0 and 300 mg/kg bw/day were treated as a recovery group. In addition, 5 females/dose 0 and 300 mg/kg bw/day groups were dosed for 42 days without mating and examined after the recovery period. At 300 mg/kg bw/day, increased salivation and decreased body weight gain were observed in both sexes. Absolute and relative kidney weights increased and hyaline droplet formation in the proximal tubular epithelium increased in males administered 300 mg/kg bw/day. Hyperplasia of the urinary bladder mucosal epithelium was observed in males administered 60 and 300 mg/kg bw/day and in females administered 300 mg/kg bw/day. Among these changes, increased relative kidney weight in males and hyperplasia of the urinary bladder mucosal epithelium in females persisted after the recovery period. Based on these effects in the kidney and urinary bladder, the NOAELs for repeated dose toxicity were determined to be 10 mg/kg bw/day and 60 mg/kg bw/day in male and female rats, respectively. |
|
UUID
|
IUC5-6d4f5ddf-6d29-41e4-b901-cef79aa25950
| |
|
Dossier
UUID |
0
| |
|
Author
|
dra /
National Institute of Health Sciences / Tokyo / Japan | |
|
Date
|
2017-01-04
16:42:56 JST | |
|
Remarks
|
|
Purpose
flag |
key study
| ||
|
Study
result type |
experimental result
|
||
|
Reliability
|
1
(reliable without restriction) | ||
|
Rationale
for reliability incl. deficiencies |
OECD Test
Guideline study under GLP condition | ||
|
Reference
type |
Author
|
Year
|
Title
|
Bibliographic
source |
Testing
laboratory |
Report
no. |
Owner
company |
Company
study no. |
Report
date |
|
study report |
MHLW (Ministry of Health, Labour and Welfare), Japan |
2006 |
Reverse Mutation Test of chlorocyclohexane on Bacteria. |
Japan Existing Chemical Data Base (JECDB) |
Mitsubishi Safety Institute Ltd. |
B041795 |
2006-09-14 |
data published |
gene mutation |
bacterial reverse mutation assay (e.g. Ames test)
|
|
Qualifier
|
Guideline
|
Deviations
|
|
according to |
OECD Guideline 471 (Bacterial Reverse Mutation Assay) |
no |
|
according to |
JAPAN: Guidelines for Screening Mutagenicity Testing Of Chemicals
|
no |
yes |
yes |
|
Identifier
|
Identity
|
|
CAS number |
542-18-7 |
- Name of test material (as cited in study report):
Chlorocyclohexane - CAS No.: 542-18-7 - Molecular weight: 118.61 - Lot No.: 5C1114 - Purity: 99.7% - Supplier: Junsei Chemical Co., Ltd. - Vapor pressure: 6.73 mmHg (25°C) - Vapor density : 4.12 (air = 1) - Boiling point : 142°C - Melting point: -44°C - Flash point: °C - Specific gravity: 1.004 (20/4°C) - Solubility: Insoluble in water and miscible by organic solvents as the alcohol and ether et al. - Odor: Characteristic odor - Physical state: Slightly pale yellow from colorless liquid - Storage condition of test material: Dark and sealed in refrigerator (2.8 - 8.4°C) |
Species/strain
|
S. typhimurium TA 1535, TA 1537, TA 98, TA 100 and E. coli WP2
|
Metabolic
activation |
with and without |
Metabolic
activation system |
SD male rat liver, induced by phenobarbital and 5,6-benzoflavone
|
-S9 mix: 2.44, 4.88, 9.77, 19.5, 39.1, 78.1, 156
μg/plate (TA100, TA1535 strains), 9.77, 19.5, 39.1, 78.1, 156, 313 μg/plate (TA98, TA1537, WP2uvrA/pKM101 strains) +S9 mix: 9.77, 19.5, 39.1, 78.1, 156, 313 μg/plate (TA100, TA1535, TA98, TA1537 strains), 9.77, 19.5, 39.1, 78.1, 156, 313, 625 μg/plate (WP2uvrA/pKM101 strain) |
- Vehicle(s)/solvent(s) used: DMSO
|
Negative
controls |
no | |
Solvent
/ vehicle controls |
yes | |
True
negative controls |
no | |
Positive
controls |
yes | |
Positive
control substance |
|
METHOD OF APPLICATION: Preincubation DURATION-
Preincubation period: 20 min at 37°C - Exposure duration:48 hrs NUMBER OF
PLATES: 3 NUMBER OF REPLICATIONS: 1 DETERMINATION OF CYTOTOXICITY- Method:
other: growth inhibition |
In any strain(s) tested with or without S9 mix, when the
mean number of revertant colonies per plate increased twice more than that
of the negative control and when the increase was shown to be dose-related
and reproducible, the chemical was judged mutagenic.
|
No |
Species/strain
|
S. typhimurium TA 1535, TA 1537, TA 98, TA 100 and E. coli WP2
|
Metabolic
activation |
with and without |
Test
system |
all strains/cell types tested |
Genotoxicity
|
negative |
Cytotoxicity
|
yes |
Vehicle
controls valid |
yes |
Negative
controls valid |
not examined |
Positive
controls valid |
yes |
RANGE-FINDING/SCREENING STUDIES: Concentration: 1.22,
4.88, 19.5, 78.1, 313, 1250, 5000 μg/plate with and without S9
mix Cytotoxic conc.: -S9 at 78.1μg/plate and higher, +S9 at 313 μg/plate and higher |
Figures and Tables (in Japanese) are available in the following full report of the study. http://dra4.nihs.go.jp/mhlw_data/home/pdf/PDF542-18-7e.pdf |
No increase in revertant colonies was observed in the test with either the non-activation method (-S9) or activation (+S9) method. Reverse mutation assays using microorganisms (Salmonella typhimurium, Escherichia coli) were conducted to assess the potential of chlorocyclohexane to induce gene mutations. Chlorocyclohexane did not induce gene mutations in the bacteria under the conditions of this study. The positive control showed expected results. |
Chlorocyclohexane did not induce gene mutations in the
in vitro bacteria test. |
|
UUID
|
IUC5-5c6ddd27-d102-4a29-91ea-e87ededc3b69
| |
|
Dossier
UUID |
0
| |
|
Author
|
dra /
National Institute of Health Sciences / Tokyo / Japan | |
|
Date
|
2017-01-04
16:43:02 JST | |
|
Remarks
|
|
Purpose
flag |
key study
| ||
|
Study
result type |
experimental result
|
||
|
Reliability
|
1
(reliable without restriction) | ||
|
Rationale
for reliability incl. deficiencies |
OECD Test
Guideline study under GLP condition | ||
|
Reference
type |
Author
|
Year
|
Title
|
Bibliographic
source |
Testing
laboratory |
Report
no. |
Owner
company |
Company
study no. |
Report
date |
|
study report |
MHLW (Ministry of Health, Labour and Welfare), Japan |
2006 |
In Vitro Chromosomal Aberration Test of Chlorocyclohexane on Cultured
Chinese Hamster Cells |
Japan Existing Chemical Data Base (JECDB) |
Mitsubishi Safety Institute Ltd. |
B041796 |
2016-09-15 |
data published |
chromosome aberration |
in vitro mammalian chromosome aberration test
|
|
Qualifier
|
Guideline
|
Deviations
|
|
according to |
OECD Guideline 473 (In vitro Mammalian Chromosome Aberration Test)
|
no |
|
according to |
JAPAN: Guidelines for Screening Mutagenicity Testing Of Chemicals
|
no |
yes |
yes |
|
Identifier
|
Identity
|
|
CAS number |
542-18-7 |
- Name of test material (as cited in study report):
Chlorocyclohexane - CAS No.: 542-18-7 - Molecular weight: 118.61 - Lot No.: 5C1114 - Purity: 99.7% - Supplier: Junsei Chemical Co., Ltd. - Vapor pressure: 6.73 mmHg (25°C) - Vapor density : 4.12 (air = 1) - Boiling point : 142°C - Melting point: -44°C - Flash point: °C - Specific gravity: 1.004 (20/4°C) - Solubility: Insoluble in water and miscible by organic solvents as the alcohol and ether et al. - Odor: Characteristic odor - Physical state: Slightly pale yellow from colorless liquid - Storage condition of test material: Dark and sealed in refrigerator (2.8 - 8.4°C) |
Chromosome |
Species/strain
|
other: Chinese hamster lung(CHL/IU) cells |
Metabolic
activation |
with and without |
Metabolic
activation system |
SD male rat liver, induced by phenobarbital and 5,6-benzoflavone
|
-S9 mix (short term treatment): 100, 150, 200, 250, 300,
350 ug/mL +S9 mix (short term treatment): 200, 300, 400, 450, 500 ug/mL Continuous treatment: 150, 175, 200, 225, 275 ug/mL |
- Vehicle(s)/solvent(s) used: DMSO
|
Negative
controls |
no | |
Solvent
/ vehicle controls |
yes | |
True
negative controls |
no | |
Positive
controls |
yes | |
Positive
control substance |
|
METHOD OF APPLICATION: Exposure duration: [continuous
treatment]: 24 hrs [short-term treatment]:6 hrs + 18 hr SPINDLE INHIBITOR: Colcemid STAIN: Giemsa stain for 20 min. NUMBER OF REPLICATIONS: 2 NUMBER OF CELLS EVALUATED: 200 cells / dose DETERMINATION OF CYTOTOXICITY - Method: relative total growth |
For the evaluation of the frequencies of structural
aberrations and of polyploidy induced, the following criteria, which are
usually used for chromosomal aberration testing with CHL, were employed.
Appearance incidence of cell with chromosomal aberrations:Negative(-):
less than 5%Equivocal(±): 5% or more, less than 10%Positive(+): 10% or
more |
No analyses |
Figures and Tables (in Japanese) are available in the following full report of the study. http://dra4.nihs.go.jp/mhlw_data/home/pdf/PDF542-18-7f.pdf |
Species/strain
|
other: Chinese hamster lung (CHL/IU) cells |
Metabolic
activation |
with and without |
Test
system |
strain/cell type: Chinese hamster lung (CHL/IU) cells |
Genotoxicity
|
negative |
Cytotoxicity
|
no |
Vehicle
controls valid |
yes |
Negative
controls valid |
not examined |
Positive
controls valid |
yes |
negative |
Chlorocyclohexane did not induce chromosomal aberrations
in cultured cells. |
Anin vitrochromosomal aberration test using CHL/IU cells (OECD TG 473) was negative with or without metabolic activation. |
|
UUID
|
IUC5-d811110c-8c27-46af-af04-96e78a861e27
| |
|
Dossier
UUID |
0
| |
|
Author
|
dra /
National Institute of Health Sciences / Tokyo / Japan | |
|
Date
|
2017-01-04
16:43:08 JST | |
|
Remarks
|
|
Purpose
flag |
key study
| ||
|
Study
result type |
experimental result
|
||
|
Reliability
|
1
(reliable without restriction) | ||
|
Rationale
for reliability incl. deficiencies |
OECD Test
Guideline study under GLP condition | ||
|
Reference
type |
Author
|
Year
|
Title
|
Bibliographic
source |
Testing
laboratory |
Report
no. |
Owner
company |
Company
study no. |
Report
date |
|
study report |
MHLW (Ministry of Health, Labour and Welfare), Japan |
2007 |
A combined repeated-dose/reproductive-developmental toxicity study of
chlorocyclohexane by oral administration in rats. |
Japan Existing Chemical Data Base (JECDB) |
Mitsubishi Safety Institute Ltd. |
B041794 |
2007-01-23 |
data published |
7.5. Repeated dose toxicity: oral: Repeated dose
toxicity: oral.001 |
other: combined repeated dose and reproduction /
developmental screening |
no |
|
Qualifier
|
Guideline
|
Deviations
|
|
according to |
OECD Guideline 422 (Combined Repeated Dose Toxicity Study with the
Reproduction / Developmental Toxicity Screening Test) |
no |
yes |
yes |
|
Identifier
|
Identity
|
|
CAS number |
542-18-7 |
- Name of test material (as cited in study report):
Chlorocyclohexane - CAS No.: 542-18-7 - Molecular weight: 118.61 - Lot No.: 5C1114 - Purity: 99.7% - Supplier: Junsei Chemical Co., Ltd. - Vapor pressure: 6.73 mmHg (25°C) - Vapor density : 4.12 (air = 1) - Boiling point : 142°C - Melting point: -44°C - Flash point: 29°C - Specific gravity: 1.004 (20/4°C) - Solubility: Insoluble in water and miscible with organic solvents such as the alcohol and ether etc. - Odor: Peculiar odor - Physical state: Slightly pale yellow from colorless liquid - Storage condition of test material: Dark and sealed in refrigerator (2.8 - 8.4°C) |
rat |
Crj: CD(SD) |
male/female |
TEST ANIMALS - Source: Atsugi Breeding Center, Charles River Laboratories Japan, Inc. - Age at study initiation:9 weeks of age - Weight at study initiation: 325-386 g for males and 193-231 g for females - Housing: bracket-type metallic wire-mesh cages/males and females excluding gestation and lactation periods (W 195 × D 325 × H 180 mm), and polycarbonate cage during gestation and lactation periods/females (W 265 × D 426 × H 200 mm) - Diet (e.g. ad libitum): ad libitum - Water (e.g. ad libitum): ad libitum excluding collected fresh urine - Acclimation period:8 days ENVIRONMENTAL CONDITIONS - Temperature (°C):21.4 to 23.7°C - Humidity (%): 48.8 to 65.0% - Air changes (per hr): 9 to 20 times per hour - Photoperiod (hrs dark / hrs light):12-hour lighting per day |
oral: gavage |
olive oil |
PREPARATION OF DOSING SOLUTIONS: Test substance was
dissolved in olive oil for injection. VEHICLE - Justification for use and choice of vehicle: No data - Amount of vehicle (if gavage): 5 ml/kg bw - Lot/batch no. (if required): No data - Dosing volume: 5 mL/kg - Stability (test solutions): At least 7 days - Storage condition of test solution: Stored in a refrigerator |
yes |
Test suspensions at each concentration of initial and
final preparations were analyzed by the GC method at Mitsubishi Safety
Institute Ltd. Results showed that the concentration of the test article
in each suspension was 95.0 to 106.2% of the nominal concentration and
both values were within the acceptable range (concentration: percentage of
the nominal concentration, 100 ± 10%) |
(P) Males: 42 days including 14 days pre-mating and
mating periods (P) Females: Days including 14 days pre-mating, mating and
gestation periods and the days until day 4 of lactation
|
Daily: 7 times / week |
0 (vehicle), 10, 60 and 300 mg/kg bw/day | |
Basis
|
actual ingested |
12 females/dose (0, 10, 60, 300 mg/kg), 7, 12, 12, 7
males of 0, 10, 60, 300 mg/kg, respectively, 5 males and 5 females at 0
and 300 mg/kg bw/day (recovery group) |
yes, concurrent vehicle |
- Dose selection rationale: A preliminary study was
conducted to determine the doses to be employed. Three males and three
female SD rats were receiving 0, 30, 100, 300, and 1000 mg/kg groups of
the substance were administered for 14 days. As a result, death or dying
was observed in all males and females receiving 1000 mg/kg groups.
Salivation and increases in absolute and relative adrenal weights were
observed in females receiving 300 mg/kg group. No clear changes related to
the test substance were observed in males receiving 300 mg/kg group.
Therefore, the high dose was set at 300 mg/kg/day, and the middle and low
dose were set at 60 and 10 mg/kg/day using common ratio 5. Vehicle control
groups were set using olive oil only. |
CAGE SIDE OBSERVATIONS: Yes - Time schedule: Males and females: once before the start of administration, two times/day during the administration period, and once during the recovery period DETAILED CLINICAL OBSERVATIONS: Yes - Time schedule: Males: once before the start of administration, during the administration and recovery periods Females: once before the start of administration, days 1, 7, 14 and 20 of gestation, and day 4 of lactation BODY WEIGHT: Yes - Time schedule for examinations: Males in the main and recovery groups were weighed on Day 1, 8, 15, 22, 29, 36, 42, and 43 of administration, and males of recovery groups were weighed on Day 50 and 56. Female satellite groups were weighted same frequencies to male recovery groups. Females in the main groups were weighed on Day 1, 8 and 15 of administration and copulated females were weighed on Day 0, 7, 14 and 20 of gestation, and days 0 and 4 of lactation. FOOD CONSUMPTION: Yes - Food consumption (g/day/rat) for each animal determined from the difference of the of the previous day's feeding amount: Yes Measurement of food consumption was conducted on all animals at the following frequencies: Males in the main and recovery groups were measured on Day 1-8, 8-15, 15-22, 22-29, 29-36, 36-38, 43-50, and 50-52. Female satellite groups were measured on Day 1-8, 8-15, 15-22, 22-29, 29-36, 36-42, 43-50, and 50-56. Main females were measured same frequencies to body weighted days. It is not measured during the mating. FOOD INTAKE: No COMPOUND INTAKE: No FOOD EFFICIENCY: No WATER CONSUMPTION: No |
Vaginal smears were collected from all females in the
main groups and microscopically examined every day from the day after the
start of administration until the day copulation was confirmed. Mean
estrous cycle (day) and abnormal estrous cycle animals (not 4 to 6 day in
estrous cycle) were examined by dams. |
Parameters examined in P male parental generations:
testes weight, epididymides weight |
PARAMETERS EXAMINED:The following parameters were
examined in F1 offspring: Number and sex of pups, stillbirths, live
births, postnatal mortality, presence of gross anomalies, and weight gain.
|
SACRIFICE: The F1 pups were euthanized on PND 4 by
exsanguination pentobarbital sodium anesthesia,
intraperitoneally. GROSS NECROPSY: Yes |
Parametric data such as grip strength, motor activity,
body weight and gain, food consumption, urine volume, specific gravity,
Hematology, blood biochemistry, and absolute and relative organ weights
were analyzed by Bartlett’s test for homogeneity of distribution. When
homogeneity was recognized, one-way analysis of variance was performed.
When a significant difference was observed, Dunnett’s multiple comparison
test was conducted for comparison between control and treated groups. If
not homogenous, analysis was performed using the Kruskal-Wallis ranking
test. In consequence, if not homogenous, Dunnett’s type mean rank sum test
was conducted to compare to control and individual treatment groups.
Qualitative value as the pathological findings was analyzed by Wilcoxon
test and Fisher’s exact test. Urinalyses data were analyzed by
Kruskal-Wallis and Dunnet’s type mean rank test. Reproductive incidences
of estrous cycle, fertility index, copulation index, delivery index, sex
ratio, and external abnormalities were analysed by Fisher’s exact test.
Significance level was set at 0.05 compared with the control group and
among the groups. |
1) Each parameter was determined by the following
equations: Mean estrus cycle, incidence of females with irregular estrus cycle, mating periods, Copulation index (%) = (No. of copulated animals/No. of co-housed animals) × 100 Fertility index (%) = (No. of pregnant females/No. of copulated females) × 100 Gestation length, number of corpora lutea, number of implantation sites, total number of offspring, Implantation index (%) = (No. of implantation sites/No. of corpora lutea) × 100 Delivery index (%) = (No. of females delivered liveborn pups/No. of pregnant females) × 100 Gestation index (%) = (No. of pregnant animals delivered live offspring/number of pregnant animals) × 100 |
Total number of offspring at birth, number of live
offspring at birth, Number of live pups on day 0 of lactationBirth index (%) = (Number of live pups on day 0/Number of implantation sites) ×100 Viability index = (Number of live pups on day 4 after birth/Number of live pups born) ×100 External examination of offspring, necropsy finding Pups weight on day 0 of lactation Sex ratio on day 0 of lactation Number of live pups on day 4 of lactation Pups weight on day 4 of lactation Sex ratio on day 4 of lactation |
|
Endpoint
|
Generation
|
Sex
|
Effect
level |
Based on
|
Basis
for effect level / Remarks |
|
NOAEL |
P |
male/female |
300 mg/kg bw/day |
No reproductive effects up to highest dose tested | |
|
NOAEL |
F1 |
male/female |
300 mg/kg bw/day |
No developmental effects up to highest dose tested
|
yes |
yes |
no effects |
no effects |
not examined |
no effects |
yes |
yes |
yes |
1) Estrous Cycle There were no animals showing abnormal estrous cycles, and there were no significant differences in the average length of the estrous cycle between the control group and any treatment group. 2) Results of Mating There were no significant differences in the incidence of females with irregular estrus cycle, mating period with the number of estrus and day of conceiving, copulation index, and fertility index between the control group and any treatment groups. 3) Delivery Data and Delivery There were no significant differences in the gestation length, number of corpora lutea, number of implantation sites, implantation index, and delivery index between the control group and any treatment groups. GROSS PATHOLOGY See 7.5.1 Repeated dose toxicity: oral HISTOPATHOLOGY See 7.5.1 Repeated dose toxicity: oral |
no effects |
no effects |
no effects |
no effects |
no data |
no effects |
no data |
Figures and Tables (in English) are available in the following full report of the study. http://dra4.nihs.go.jp/mhlw_data/home/pdf/PDF542-18-7d.pdf |
The NOAEL for rat reproductive and developmental
toxicity was determined to be 300 mg/kg bw/day.
|
A combined repeated oral dose toxicity study and reproduction/developmental toxicity screening test was performed according to the OECD TG 422. Male and female rats (12 animals/sex/dose) were administered chlorocyclohexane at 0, 10, 60, and 300 mg/kg bw/day. Males were dosed for 42 days, including a 14 day pre-mating period and subsequent mating period. Females were dosed for up to 55 days, including 14 day pre-mating, mating, and gestation periods, and the time until lactation day 4. Five out of 12 males dosed at 0 and 300 mg/kg bw/day were treated as a recovery group. In addition, 5 females/dose 0 and 300 mg/kg bw/day groups were dosed for 42 days without mating and examined after the recovery period. There were no effects on reproductive and developmental parameters at 300 mg/kg bw/day. The NOAEL for the rat reproductive/developmental toxicity of chlorocyclohexane was determined to be 300 mg/kg bw/day, the highest dose tested. |
|
UUID
|
ECB5-9483ca56-6e33-4ec9-a0f4-545b621d4c14
| |
|
Dossier
UUID |
0
| |
|
Author
|
XML
Transformation V4.0 Plug-In | |
|
Date
|
2007-05-10
18:00:00 JST | |
|
Remarks
|
Successfully
migrated to IUCLID 5.5 format. |
|
Reference
substance name |
chlorocyclohexane
|
|
EC
number |
208-806-6 |
CAS
number |
542-18-7 |
|
EC
name |
chlorocyclohexane
| ||
|
Molecular
formula |
C6H11Cl
| ||
|
CAS
number |
542-18-7
|
|
chlorocyclohexane
|
|
Name
|
chlorocyclohexane
|
|
Name
|
Cyclohexane, chloro-
|
|
Name
|
Cyclohexane, chloro-
|
|
DSL Category: Organics
|
|
Molecular
formula |
C6H11Cl |
|
Molecular
weight range |
118.6045 |
|
SMILES
notation |
ClC1CCCCC1
|
|
InChI
|
InChI=1/C6H11Cl/c7-6-4-2-1-3-5-6/h6H,1-5H2
|
|
Structural
formula |
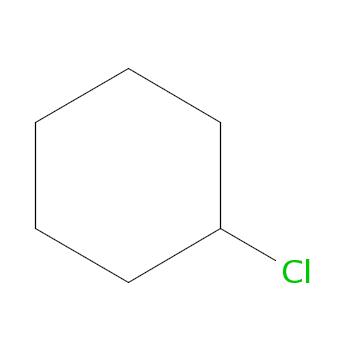 |
|
UUID
|
IUC4-b036ff75-0f3c-323b-b200-ed5f46cf5101
| |
|
Dossier
UUID |
0
| |
|
Author
|
XML
Transformation V4.0 Plug-In | |
|
Date
|
2011-06-23
11:55:01 JST | |
|
Remarks
|
Successfully
migrated to IUCLID 5.5 format. |
|
Legal
entity name |
National Institute of
Health Sciences |
|
Flags |
IT system |
ID |
Remarks |
|
LEO |
10767 |
||
|
IUCLID4 |
16558402024DIV750
|
|
Address
|
1-18-1 kamiyoga
|
|
Address
|
Setagaya-ku
|
|
Postal
code |
158-8501 |
|
Town
|
Tokyo |
|
Country
|
Japan
|
|
Organisation
|
National Institute of
Health Sciences |
|
Department
|
Division of Risk
Assessment |
|
Title
|
Dr. |
|
First
name |
Akihiko |
|
Last
name |
Hirose |
|
Address
|
1-18-1 Kamiyoga
|
|
Address
|
Setagaya-ku
|
|
Postal
code |
158-8501 |
|
Town
|
Tokyo |
|
Country
|
Japan
|